Translate this page into:
The systematic effects of chitosan on fibroblasts derived from hypertrophic scars and keloids
Correspondence Address:
Jingde Zhang
Department of Plastic Surgery, Changhai Hospital, Second Military Medical University, 168 Changhai Road, Shanghai 200433
China
| How to cite this article: Lv C, Dai H, Xing X, Zhang J. The systematic effects of chitosan on fibroblasts derived from hypertrophic scars and keloids. Indian J Dermatol Venereol Leprol 2012;78:520 |
Sir,
Each year, millions of patients may develop scars around the globe, resulting from elective operations and operations after trauma, and the predisposed individuals would eventually develop hypertrophic scars (HTS) or keloids. [1] Nowadays, their prevention and treatment still represent a challenge for doctors. Chitosan is a functional, renewable, non-toxic and biodegradable biopolymer and is divided into 3 crystal types: α, β and γ type, among these, α-type is the most common type obtained from crust of shrimp and crab. Due to its various biological properties including biocompatibility, cationic, antitumor, immunoenhancing, antimicrobial, hemostatic properties, chitosan has been put into diverse applications, especially in pharmaceutical, food and cosmetics. In the medical field, chitosan is now used in wound healing and tissue repair in various available forms such as hydrogels, xerogels, powders, composites, films and scaffolds. [2] Compared with gauze dressings, chitosan hemostatic products could reduce blood loss and increase patient survival. [3] It is reported to enhance the recovery of venous leg ulcer in a longitudinal and descriptive study. [4] Chitosan′s properties also allow it to be used in transdermal drug delivery, such as for the transport of insulin. [5] Chitosan has also been reported to be able to reduce scar formation. [6] However, the potential mechanisms underlying the inhibitory effects of chitosan on scar formation has not been fully explored. We aimed to examine the effect of chitosan on the fibroblasts, derived from hypertrophic scar and keloids tissues in terms of proliferation, mRNA expression of type I and III procollagen and secretion of scar formation-associated cytokines including TGF-β1, bFGF and IL- 8. Fresh specimens were obtained from inpatients and outpatients in our department, 6 keloid patients and 6 hypertrophic scars patients.
The obtained specimens from patients should be maintained in Dulbecco′s modified Eagle′s medium (DMEM) (Gibco, Califórnia, USA), put on an ice and processed within 4 hours. Fibroblast harvesting is done by explants. Fibroblasts are used between the 4 th and 7 th passage for experiments. In this study, hypertrophic scar fibroblast (HSF), keloids fibroblast (KF) as well as normal dermal fibroblast (NDF) were cultured in the presence or absence of indicated dosages of chitosan (Sunfive Co., Ltd), ranging from 20 to 320 ug/ml for 72 hours. As demonstrated in [Figure - 1], 3- [4,5-dimethylthiazol-2yl]-diphenyltetrazolium bromide (MTT) assay results showed that HSF and KF had higher proliferative rates than NDF without chitosan treatment (P < 0.01), and chitosan inhibited proliferation of HSF and KF in a dose-dependent manner. Chitosan also displayed dose-dependent proliferation-inhibiting effect on NDF to a lesser extent. 320 ug/ml chitosan almost reduced the proliferation of HSF to < 70% of the control (P < 0.01). The proliferation of KF was reduced by almost 40% when treated with chitosan at 320 ug/ml (P < 0.01). Next, we examined the mRNA expression levels of type I and III procollagen in HSF and KF with or without chitosan treatment by realtime RT-PCR. The primers for type I procollagen were as follows: Forward primer 5` -TTCTGGAGCACAGCAGAAGA -3` and reverse primer 5`-TCTGCCTGCTCCATACACAC-3`, and for type III procollagen were forward primer 5`-CCCCGGTCCTAAAGGAAAT-3` and reverse primer 5`-TCTGCCTGCTCCATACACAC-3`. As demonstrated in [Figure - 2], results showed that chitosan could greatly suppress type I and III procollagen expression in both HSF and KF (P < 0.01). It is known that growth factors such as, TGF-β and bFGF play an important role in modulating wound healing and is associated with increased collagen synthesis. [1] We found that in the absence of chitosan, HSF and KF had almost doubling of the amounts of TGF-β1 and bFGF in the supernatants compared with NDF as determined by ELISA. Treatment of chitosan led to dramatic reduction of TGF-β1 and bFGF in the supernatant of HSF, KF and NDF, which was also in a dose-dependent way [Figure - 3]. Furthermore, IL-8 was reported to be able to stimulate collagenase activity in various cells as well as in HSF. [7] As shown in [Figure - 4], The baseline IL-8 production is similar in all 3 types of fibroblasts, and chitosan treatment could increase IL-8 production in a dose-dependent manner. With 320 ug/ml chitosan treatment, the amounts of IL-8 were almost as twice as those in control groups (P < 0.01).
 |
| Figure 1: Chitosan inhibits the proliferation of HSF and KF in a dose-dependent manner. MTT assay results showed that chitosan inhibited proliferation of HSF and KF in a dose-dependent manner. Data of 3 replicates (means ± SD) are shown as absorption value. (* P < 0.05; † P < 0.01 compared with their separate control groups) |
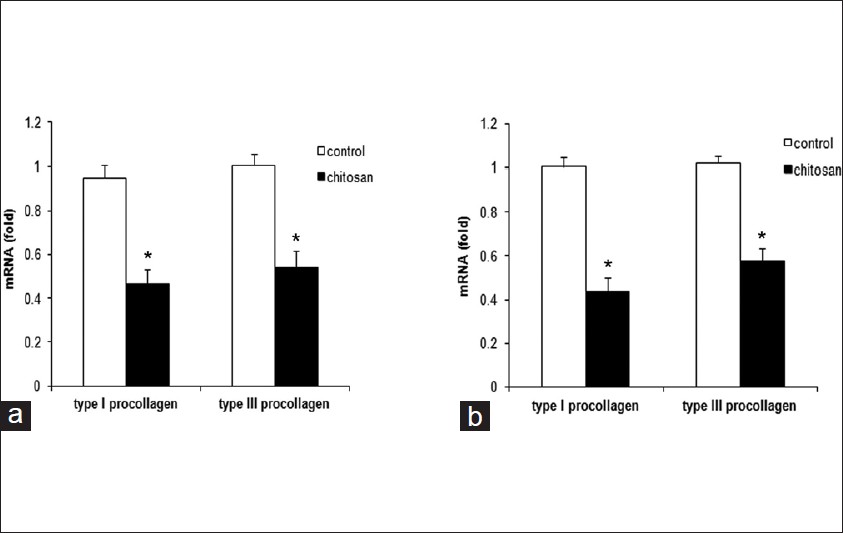 |
| Figure 2: Chitosan suppresses type I and III procollagen production by HSF and KF. RT-PCR results showed that chitosan greatly suppressed type I and III procollagen levels by HSF (a) and KF (b). GAPDH(glyceraldehyde-3-phosphate dehydrogenase) mRNA was used to normalize the variability in template loading. The data are reported as mean ± SD. (* P < 0.01) |
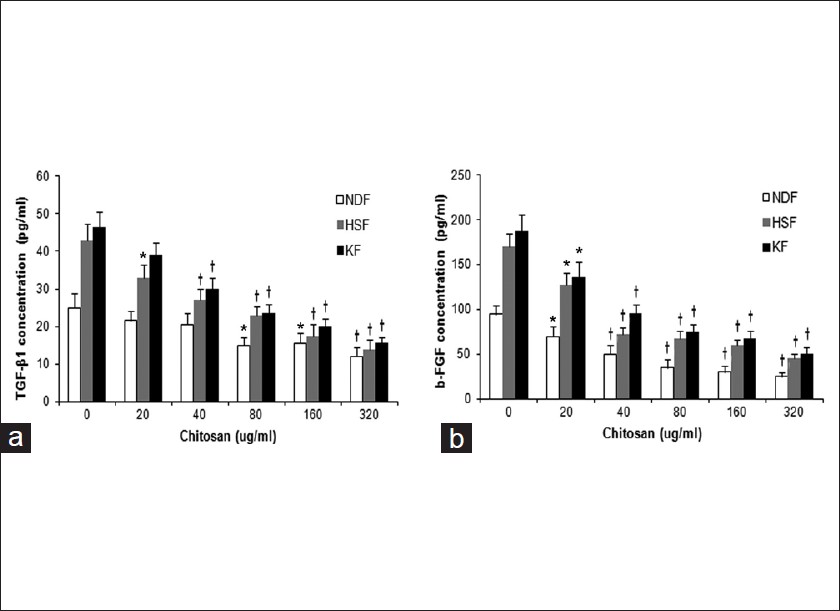 |
| Figure 3: Chitosan reduces TGF-β1 , bFGF production by HSF and KF. TGF-β1 (a) and bFGF (b) production by HSF and KF were significantly downregulated upon chitosan treatment in a dose-dependent manner as determined by ELISA. The data are reported as mean ± SD. (* P < 0.05; † P < 0.01 compared with their separate control groups) |
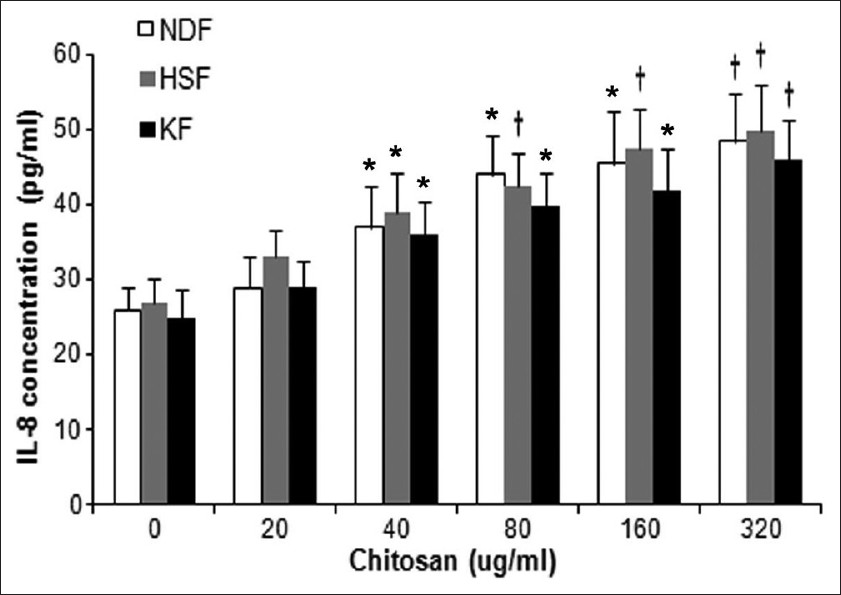 |
| Figure 4: Chitosan promotes IL-8 production by HSF and KF. IL-8 production by HSF and KF were upregulated upon chitosan treatment in a dose-dependent manner as determined by ELISA. The data are reported as mean ± SD. (* P < 0.05; † P < 0.01 compared with control group) |
Histologically, HTS and keloids result from increased fibroblast density and an overabundance of dermal collagen. [1] Thus, an inhibition of fibroblast proliferation and collagen secretion by fibroblasts is of important significance for prevention of HTS and keloids. Fibroblasts not only produce extracellular matrix (ECM) but also secrete a broad array of cytokines, which are closely involved in wound healing and scar formation process, including transforming growth factor (TGF)-β1, basic fibroblast growth factor (bFGF) and interleukin-8 (IL-8). Changes in the levels of these growth factors could lead to significant alteration in composition of the ECM. [1],[7] All of the above reveals the concept that a transition of the normal fibroblast phenotype to keloids and hypertrophic scars fibroblasts is essential for the formation of these 2 types of scars. [1] Consistently, our data showed that the HSF and KF had higher proliferative capacity and produced higher level of fibrogenic factors TGF-β1 and bFGF than NDF. Our data further revealed that chitosan showed effective inhibitory capacity for the proliferation of HSF and KF and their production of TGF-β1 and bFGF as well as type I and III procollagen. At the same time, chitosan treatment led to dramatic increase in IL-8 production by HSF and KF although the 3 types of fibroblasts had similar baseline expression of IL-8 in the culture supernatant without chitosan treatment. While our data showed that chitosan also had inhibitory effects on NDF proliferation, there are other reports showing that chitosan may stimulate the proliferation and ECM production of fibroblasts derived from normal dermis. [8],[9] The controversial findings may be attributable to different models researchers chose as well as different donor, culture medium, the dosage of chitosan, etc. Overall, our research could help to further unveil the mechanisms underlying scar prevention effect of chitosan, including inhibition of fibroblast proliferation, procollagen production as well as regulation of TGF-β, bFGF and IL-8 production, thus providing the evidence that chitosan has more potential to be used as wound healing and tissue repair agent or dressing material.
Acknowledgements
This project was supported by the Shanghai Science and Technology Committee (Grant No: 09ZR1400600).
| 1. |
Gauglitz GG, Korting HC, Pavicic T, Ruzicka T, Jeschke MG. Hypertrophic Scarring and Keloids: Pathomechanisms and Current and Emerging Treatment Strategies. Mol Med 2011;17:113-25.
[Google Scholar]
|
| 2. |
Zhang JL, Xia WS, Liu P, Cheng QY, Tahirou T, Gu WX, et al. Chitosan Modification and Pharmaceutical/Biomedical Applications. Mar Drugs 2010;8:1962-87.
[Google Scholar]
|
| 3. |
Pusateri AE, McCarthy SJ, Gregory KW, Harris RA, Cardenas L, McManus AT, et al. Effect of a chitosan-based hemostatic dressing on blood loss and survival in a model of severe venous hemorrhage and hepatic injury in swine. J Trauma 2003;4:177- 82.
[Google Scholar]
|
| 4. |
Sandoval M, Albornoz C, Muñoz S, Fica M, García-Huidobro I, Mertens R, et al. Addition of chitosan may improve the treatment efficacy of triple bandage and compression in the treatment of venous leg ulcers. J Drugs Dermatol 2011; 10:75-9.
[Google Scholar]
|
| 5. |
Agnihotri SA, Mallikarjuna NN, Aminabhavi TM. Recent advances on chitosan-based micro- and nanoparticles in drug delivery. J Control Release 2004;100:5-28.
[Google Scholar]
|
| 6. |
Chen XG, Wang Z, Liu WS, Park HJ. The effect of carboxymethyl-chitosan on proliferation and collagen secretion of normal and keloid skin fibroblasts. Biomaterials 2002;23:4609-14.
[Google Scholar]
|
| 7. |
Zhou LJ, Inoue M, Gunji H, Ono I, Kaneko F. Effects of prostaglandin E1 on cultured dermal fibroblasts from normal and hypertrophic scarred skin. J Dermatol Sci 1997;14: 217-24.
[Google Scholar]
|
| 8. |
Howling GI, Dettmar PW, Goddard PA, Hampson FC, Dornish M, Wood EJ. The effect of chitin and chitosan on the proliferation of human skin fibroblasts and keratinocytes in vitro. Biomaterials 2001;22:2959-66.
[Google Scholar]
|
| 9. |
Ueno H, Mori T, Fujinaga T. Topical formulations and wound healing applications of chitosan. Adv Drug Deliv Rev 2001;52:105-15.
[Google Scholar]
|
Fulltext Views
2,102
PDF downloads
1,289





