Translate this page into:
Topical imiquimod as a neo-adjuvant chemotherapy for cartilage salvage in the treatment of nodular basal cell carcinomas of the head and neck
Corresponding author: Dr. Saurabh Singh, Dermatology Out-Patient Department Block, All India Institute of Medical Sciences, Basni 2nd Phase, Jodhpur - 342 005, Rajasthan, India. saurabhdoc@yahoo.co.in
-
Received: ,
Accepted: ,
How to cite this article: Singh S, Rajan MB, Bhardwaj A. Topical imiquimod as a neo-adjuvant chemotherapy for cartilage salvage in the treatment of nodular basal cell carcinomas of the head and neck. Indian J Dermatol Venereol Leprol 2021;87:452-4.
Problem
Basal cell carcinomas (BCCs) on ears are categorized as high-risk, irrespective of the tumor size or histology.1 Mohs micrographic surgery remains the treatment of choice for high-risk BCCs, but the set-up is largely unavailable in developing countries like India, where wide local excisions are the most pragmatic approach. But on the ear or nose, excision often poses problems owing either to the primary destruction of cartilage by the tumor or the removal of a portion of the cartilage as part of the required 0.4–0.5 cm tumor-free margin. Naturally, such surgeries require reconstruction using grafts or local flaps, which carries the disadvantage of being expensive and time-consuming, with a protracted postoperative recovery period. Imiquimod is a promising medical therapy for small BCCs. Although imiquimod 5% cream is approved by the United States Food and Drug Administration for treatment of extra-facial, small, superficial BCCs using a 5 days/week regimen, lower quality evidence suggests that it may also be used as an alternative, albeit with lower efficacy, in small nodular BCCs which are poor candidates for surgery.2 The combination of imiquimod with surgical modalities like Mohs micrographic surgery for nodular BCC have yielded ambiguous results.3,4 In contrast, a pilot study reported encouraging results with the combination of curettage and electrodessication followed by imiquimod cream once daily for 1 month.5 However, to our knowledge, none of these treatment approaches have been reported for nodular BCCs on cartilage-rich areas like the ear helix. Another common problem in nodular BCCs is that imiquimod 5% cream produces a robust irritant reaction and thus, we have never been able to keep our patients on daily imiquimod therapy.
Solution
We propose that the nodular BCCs on cartilage-rich areas may be treated with imiquimod cream first, as a neo-adjuvant chemotherapy and the tumor remaining thereafter be dealt with minor surgical procedures like curettage. This approach can avoid extensive surgical excision and closure, especially at cartilage-rich sites like the ear helix and nose tip. The corresponding author (SS) has used this approach previously at the department of dermatology at All India Institute of Medical Sciences (AIIMS), Delhi [Figures 1a-d] and is subsequently using the same at AIIMS, Jodhpur [Figures 2a-d]. The lesion is treated initially with topical imiquimod 5% cream, 3 days/ week till a total of 12 to 16 weeks [Figures 1a, b and 2a, b]. In view of the exaggerated inflammatory response in the form of moderate edema, pustulation/ulceration or severe pain, a topical mild steroid-antibiotic combination is prescribed to be applied on imiquimod off-days. Thereafter, the residual lesion is curetted using a dermal curette (stainless-steel, sized 4 mm) with repeat multi-layered shave excision of the pigmented and nodular areas, till the bright-red dermal base is visible. It is then left to heal with secondary intention and the wound generally re-epithelizes completely in 15-20 days [Figures 1c and 2c]. We have followed up 2 patients with BCCs on ear helix treated using this approach with excellent results. The wounds healed without scarring or hyperpigmentation and have remained free of recurrence for the past 7.5 years and 2 years, respectively [Figures 1d and 2d].
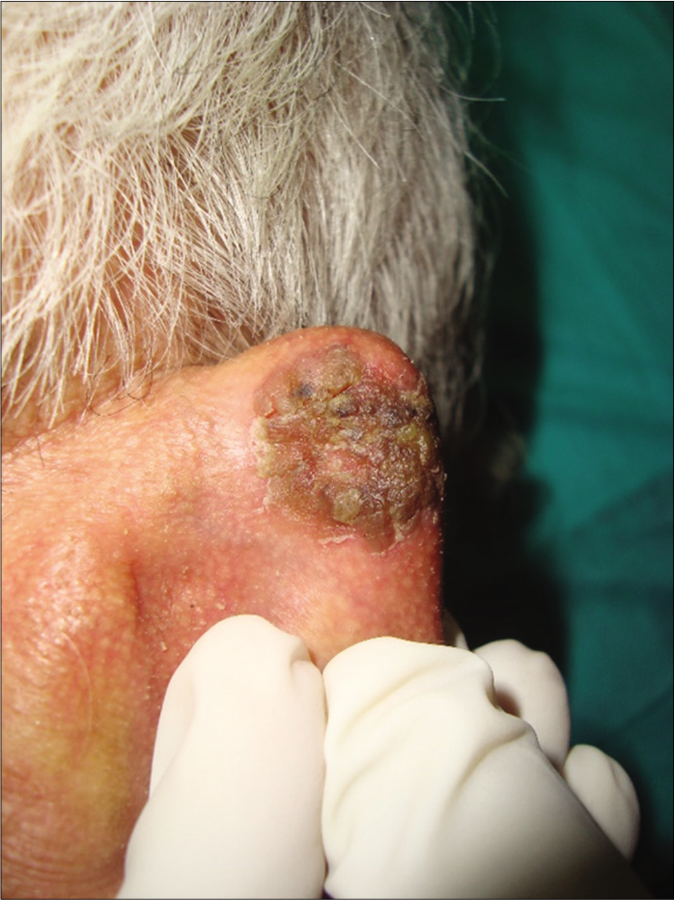
- Patient 1, nodular basal cell carcinoma on the posterior aspect of ear helix: (1.5 cm × 2 cm)
- Patient 1, nodular basal cell carcinoma on the posterior aspect of ear helix: Inflamed plaque after 12 weeks of imiquimod 5% cream application for 3 days/week
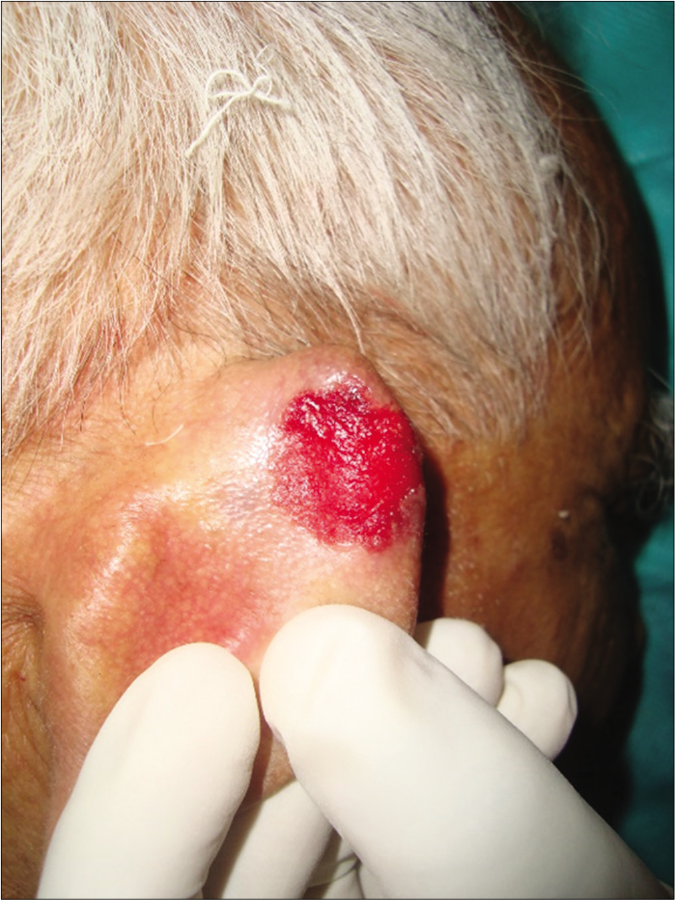
- Patient 1, nodular basal cell carcinoma on posterior aspect of ear helix: Erythematous areas after curettage till deep dermis
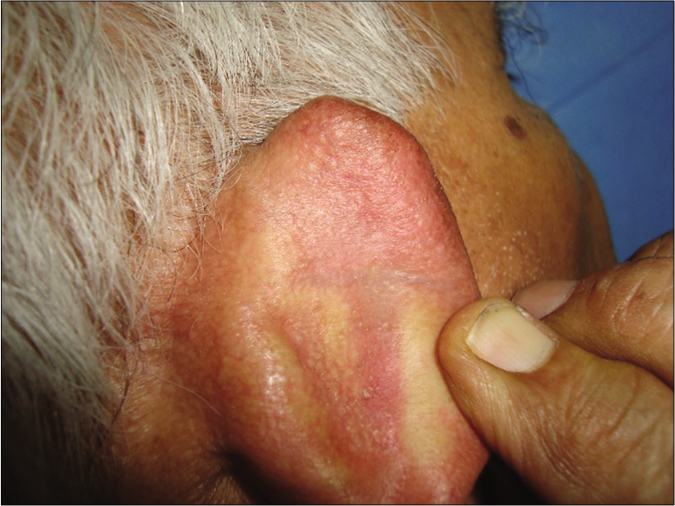
- Patient 1, nodular basal cell carcinoma on the posterior aspect of ear helix: Wound after curettage healed without any scarring/dyspigmentation at 5 years follow up.
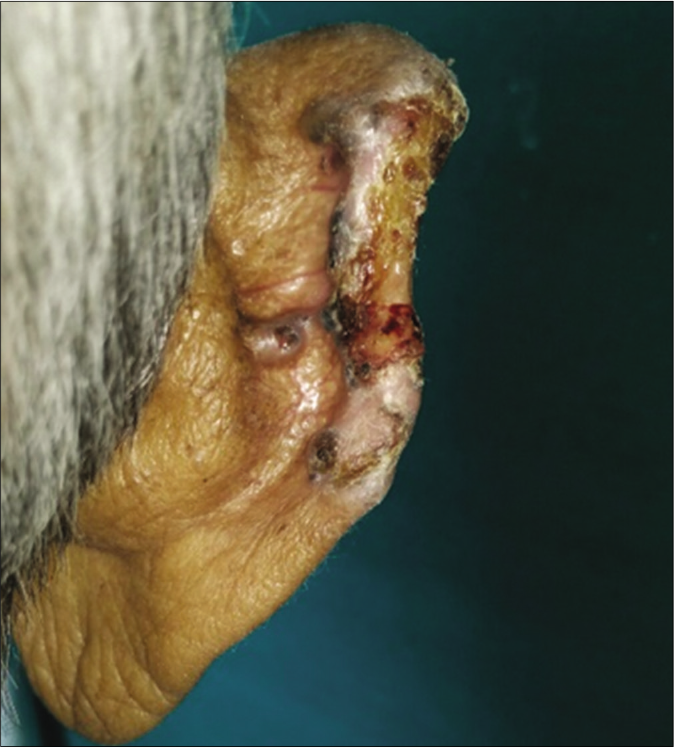
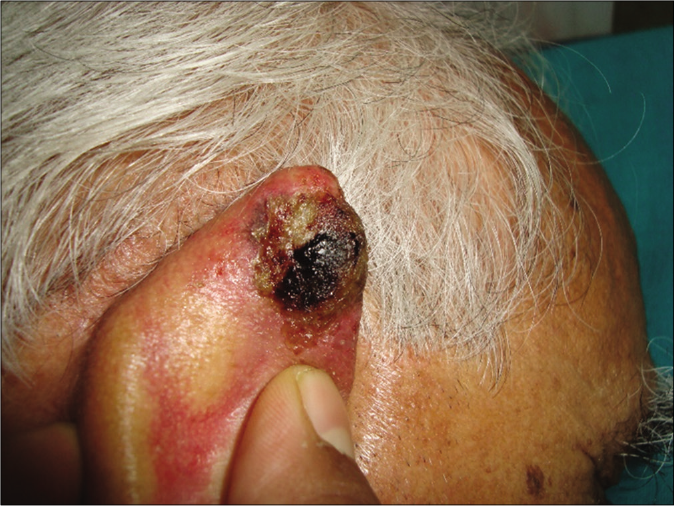
- Patient 2, noduloulcerative basal cell carcinoma on the rim of the ear: (2.5 cm × 1 cm)
- Patient 2, noduloulcerative basal cell carcinoma on the rim of ear: Residual papules after 16 weeks of imiquimod 5% cream application, 3 days/week
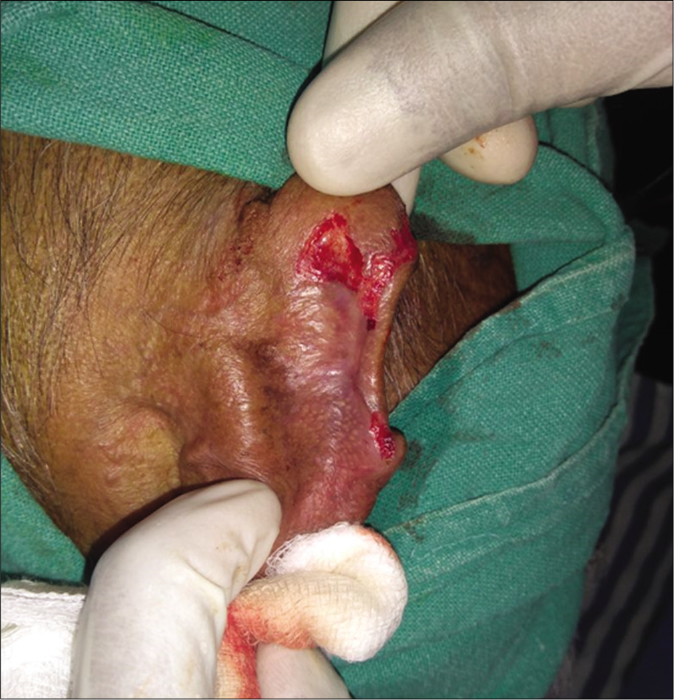
- Patient 2, noduloulcerative basal cell carcinoma on the rim of ear: Erythematous areas after curettage till deep dermis
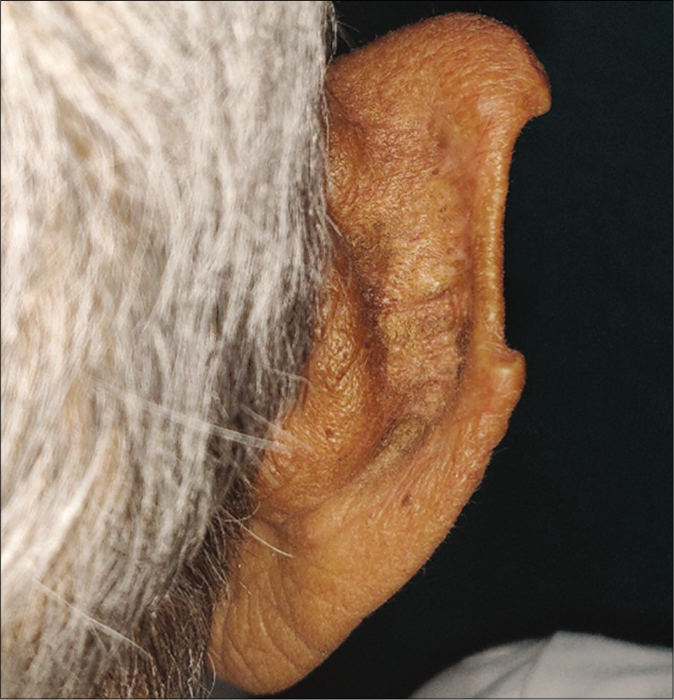
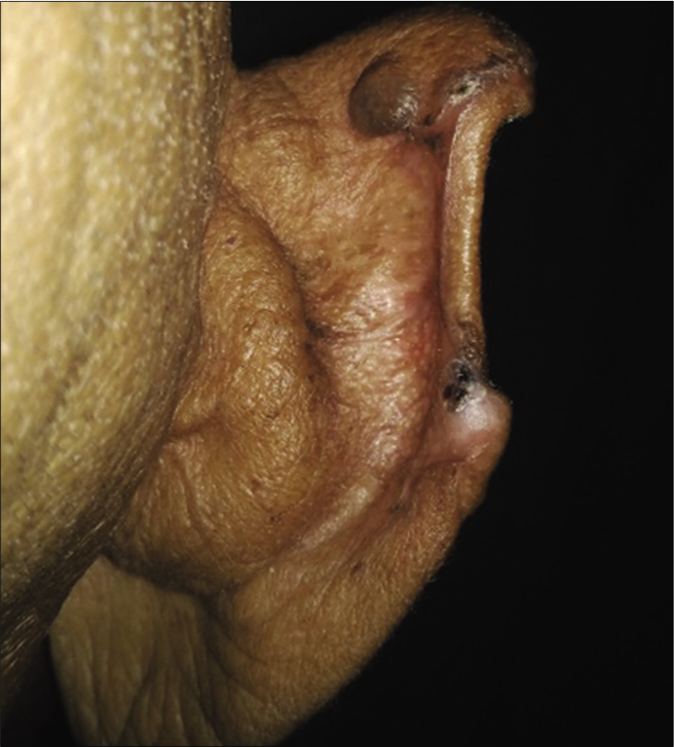
- Patient 2, noduloulcerative basal cell carcinoma on the rim of ear: Wound healed without scarring/dyspigmentation at 20 months follow up
Our approach draws inspiration from the neoadjuvant chemotherapy for solid organ malignancies requiring surgery wherein chemotherapy reduces the tumor size and improves outcome of subsequent surgical excision. Our regimen of using an alternate day imiquimod protocol followed by curettage is especially useful since it is cheaper, more tolerable and requires minimal surgical expertise. Therefore, it could be more easily adapted by dermato-surgeons.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Guidelines of care for the management of basal cell carcinoma. J Am Acad Dermatol. 2018;78:540-59.
- [CrossRef] [PubMed] [Google Scholar]
- Topical pharmacotherapy for skin cancer: Part II: Clinical applications. J Am Acad Dermatol. 2014;70:979.e1-12.
- [CrossRef] [PubMed] [Google Scholar]
- Imiquimod 5% cream as adjunctive therapy for primary primary, solitary, nodular nasal basal cell carcinomas before Moh's micrographic surgery: A randomized, double blind, vehicle-controlled study. Dermatol Surg. 2009;35:24-9.
- [CrossRef] [PubMed] [Google Scholar]
- Imiquimod 5% cream as pretreatment of Moh's micrographic surgery for nodular basal cell carcinoma in the face: A prospective randomized controlled study. Br J Dermatol. 2012;167:110-5.
- [CrossRef] [PubMed] [Google Scholar]
- Pilot study of 5% imiquimod cream as adjunctive therapy to curettage and electrodessication for nodular basal cell carcinoma. Dermatol Surg. 2006;32:63-9.
- [CrossRef] [PubMed] [Google Scholar]





