Translate this page into:
Ultrathin split-thickness skin graft template using antiseptic tulle gras dressing for vitiligo
Corresponding author: Dr. Somesh Gupta, Department of Dermatology and Venereology, All India Institute of Medical Sciences, New Delhi, India. someshgupta@hotmail.com
-
Received: ,
Accepted: ,
How to cite this article: Sindhuja T, Chandra AD, Gupta S. Ultrathin split-thickness skin graft template using antiseptic tulle gras dressing for vitiligo. Indian J Dermatol Venereol Leprol 2023;89:785
Problem
Ultrathin split-thickness skin grafting is an effective surgical procedure for treating recalcitrant vitiligo that provides rapid repigmentation.1 Ultrathin grafts in vitiligo have minimal dermal tissue and are shed after 1–2 weeks following transfer of melanocytes to the patch. Ultrathin grafts are fragile and may tear on undue manipulation. These grafts should be confined to the dermabraded recipient site and 2–3 mm beyond, as their extension into the normal skin carries a risk of displacement.
Solution
We propose using antiseptic tulle gras dressing (Bactigras, Smith & Nephew, Hull, United Kingdom) (alternatively, a paraffin gauze) to prepare a template matched with the shape and size of the recipient patch. The depigmented area with 2 mm normal surrounding skin is outlined using a surgical pen [Figure 1]. The marked boundary is traced onto the translucent tulle. The graft is placed on the tulle after removing the butter paper cover on one side, keeping the epidermal surface of the graft in contact with the tulle [Video 1]. After spreading the graft evenly, the graft-dressing composite is trimmed along the marking. The graft-tulle template [Figure 2] is placed on the dermabraded recipient site with the dermal surface facing down. After removing the outer butter paper, the graft is secured with gauze and Tegaderm (3M, St Paul).
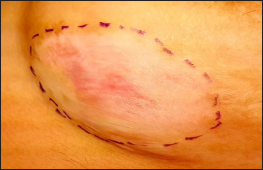
- Delineated recipient site in a patient with stable segmental vitiligo

- Ultrathin split-thickness skin graft- tulle gras template
The graft-tulle template offers an effortless way to apply the graft over the recipient site directly. It prevents wrinkling and curling of the edges and graft displacement. Pre-defining the recipient and graft areas help prevent peripheral depigmented halo due to graft shrinkage. The tulle gras dressing, which is cut to match the recipient area, can also be used to transfer the donor tissue in other techniques like suction blister grafting, non-cultured epidermal suspension and the Jodhpur technique.
Declaration of patient consent
Patient’s consent not required as patient’s identity is not disclosed or compromised.
Financial support and sponsorship
Nil.
Conflict of interest
There are no conflicts of interest.
References
- Thin and ultrathin skin grafts for vitiligo In: Gupta S, Olsson MJ, Parsad D, Lim HW, Van Geel N, Pandya AG, eds. Vitiligo: Medical and Surgical Management (1st ed.). Oxford: Wiley–Blackwell; 2018. p. :241-48.
- [Google Scholar]





