Translate this page into:
Verruciform xanthoma of the thumb
2 Department of Pathology, Gulou Affiliated Hospital to Medical College of Nanjing University, Nanjing 210008, People's Republic of China
Correspondence Address:
Yihua Wang
Department of Pathology, Gulou Affiliated Hospital of Nanjing University, No. 321, Zhongshan Road, Nanjing 210008
People's Republic of China
| How to cite this article: Li Z, Wang Y. Verruciform xanthoma of the thumb. Indian J Dermatol Venereol Leprol 2018;84:67-69 |
Sir,
Verruciform xanthoma is a rare lesion with a predilection for oral mucosa. Recent reports revealed that it affected the skin and mucosa of parts other than the oral cavity, such as that of the penis and scrotum. We present a case of verruciform xanthoma involving the thumb.
An 80-year-old woman consulted our department for an asymptomatic lesion on her left thumb, which had gradually enlarged over the past 3 years. The patient had pared the lesion with a knife by herself several times; however, it recurred every time. The lesion was diagnosed as verruca vulgaris and frozen three times by liquid nitrogen, which was not effective. She had no known history of trauma or infection of the thumb. Physical examination revealed a single ill-demarcated yellow-tan papule 0.8 cm × 0.8 cm in diameter, on the skin of the left thumb [Figure - 1]. The personal and familial medical histories of the patient were unremarkable, and no laboratory findings suggestive of hyperlipidemia were found.
 |
| Figure 1: A verrucous papule on the left thumb |
The lesion was surgically removed and sent to pathology department for histopathological examination. Hematoxylin–eosin staining revealed a hard verrucous lesion, and the histopathology showed the epidermal hyperkeratosis and parakeratosis, and a papillomatous proliferation without atypia. The papillary dermis was filled with an infiltrate of lymphocytes and plasma cells; some foamy cells were observed in it [Figure - 2] and [Figure - 3]. But the foamy cells did not extend beyond the tips of the rete ridges. The immunohistochemical examination revealed positive staining of the foamy cells for CD68 [Figure - 4], CD163, CD45, and CD3, and negative staining for CD1a, CD117, EMA, CD20, and S100. Also, the lymphocytes in the dermis stained positively with CD45 and CD20, but were negative for CD3. The epidermis was extensively positive for cytokeratin (CK), but the positive pattern of the foamy cells to the same was punctiform. The foamy cells contained scanty periodic–acid Schiff (PAS)-positive diastase-resistant material and were negative for Giemsa (GMS) staining. The patient's histopathological diagnosis was established as verruciform xanthoma, and no recurrence had occurred at 8 months follow-up.
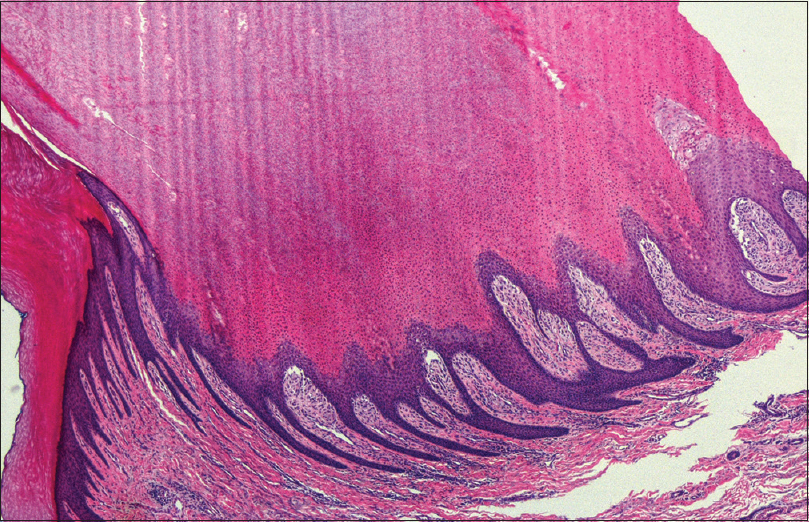 |
| Figure 2: Hematoxylin–eosin (H and E) staining revealed a hard verrucous lesion, with the epidermal hyperkeratosis, parakeratosis, acanthosis, and a papillomatous proliferation. Atypia of epithelial cells was not present (H and E, ×40) |
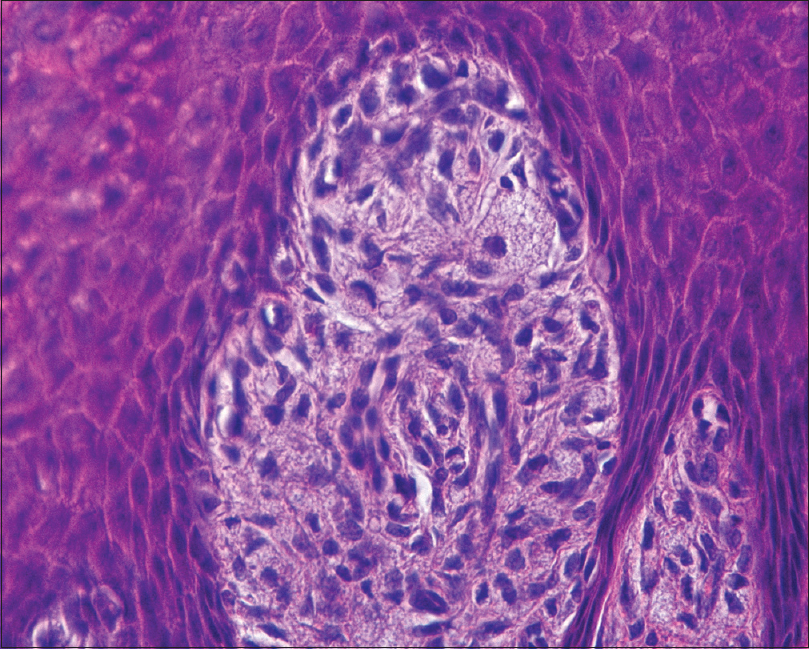 |
| Figure 3: The papillary dermis between the acanthotic rete ridges was filled with an infiltrate of lymphocytes and plasma cells. And some foamy cells were observed in the papillary dermis (H and E, ×400) |
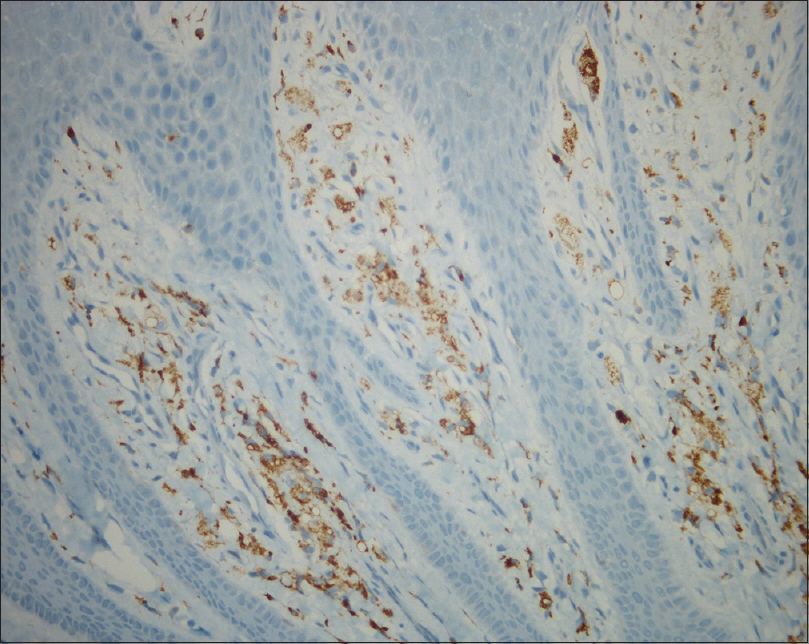 |
| Figure 4: The foamy cells are positive for CD68 ×200, a macrophage marker |
The histopathological pattern of our patient's lesion is identical to that of a typical verruciform xanthoma. It was generally accepted that this condition was first described by Shafer in 1971,[1] but Mario declared that the lesion of verruciform xanthoma had been reported by Otto in 1903.[2] It usually presents as a solitary, sessile, or pedunculated lesion with rough or pebbly surface,[3] and it may present as papules, nodules, or plaques. The lesion is generally asymptomatic, and it is about 2 mm to 2 cm in size with variable color, rarely bigger than 4 cm. Although verruciform xanthoma can occur at any age, it usually affects adults between 40 and 70 years with a slight male predilection. Oral verruciform xanthoma is common in males below the age of 50 years, and a reverse trend was noted in females.[4]
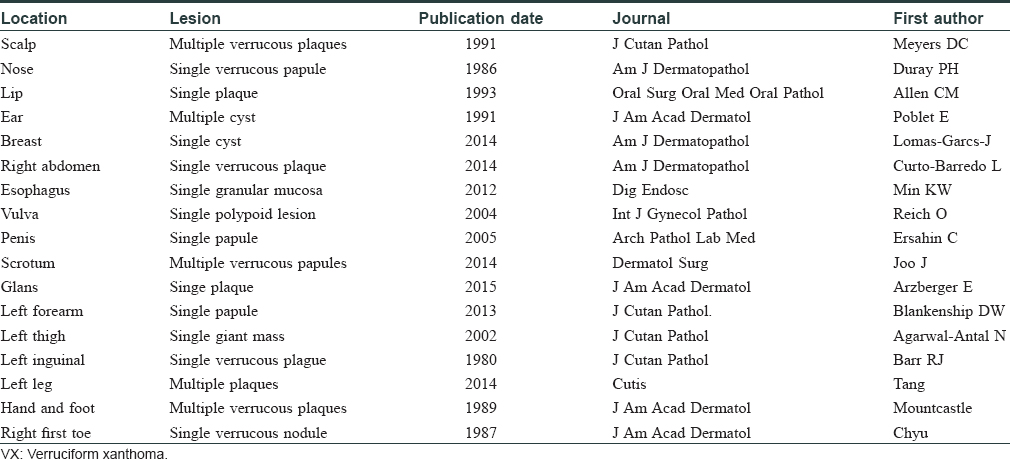
Verruciform xanthoma occurs as an isolated solitary lesion mostly. Multiple lesions were reported to be associated with other skin diseases or oral mucosal diseases, such as snuff dipper's keratosis, oral pemphigus vulgaris, carcinoma in situ, lichen sclerosus, solar keratoses, epithelial nevus, CHILD syndrome, etc.[3] Also, multiple lesions have been reported to occur in an immunocompromised patient and in hepatitis C virus carriers.[5] Verruciform xanthoma usually occurs intraorally, and cutaneous involvement is unusual. The extraoral occurrence has been reported mainly involving the anogenital skin and mucosa. There are various reports about verruciform xanthoma in other anatomic sites, for example, scalp, breast, thigh, etc. Some reports about verruciform xanthoma in extraoral sites are shown in [Table - 1]. We had found several cases of verruciform xanthoma in the leg, finger, and toe.[6],[7],[8] One of the cases was reported by Mountcastle in 1989,[8] and the lesions were in the left third digit and right toes.
The diagnosis of verruciform xanthoma is based on the histopathological examination. It is characterized by papillomatous acanthosis, aggregations of xanthoma cells located within the papillary dermis, accompanied by infiltration of lymphocytes. The histopathological pattern of our patients was atypical to that, and the foamy cells were suggested to be monocyte-macrophage lineage by immunohistochemical staining. The negativity of foamy cells to S-100 ruled out the original possibility of them originating from dermal dendritic cells. The xanthoma cells are large cells with foamy cytoplasm. They stained positively with periodic acid Schiff and are diastase-resistant, implying that the material in the foamy cells is not glycogen.
The etiology of verruciform xanthoma is not fully characterized, and it did not seem to be related to any lipid metabolism abnormality.[3] It was suggested that verruciform xanthoma is a reaction pattern to an inciting event of epidermal keratinocyte damage. Keratinocyte damage has been attributed to a wide variety of instigators including recurrent cutaneous irritation or trauma, bacterial colonization, fungi, virus, and other inflammatory damage. The keratinocyte necrosis and degeneration may lead to the release of intracellular lipids, which are then engulfed by histiocytes in the dermis, giving rise to foamy cells. Moreover, inflammation was considered to be the primary trigger. In our patient, the positive pattern of cytokeratin staining for the foamy cells confirmed this opinion. Mehra et al. studied nine cases of verruciform xanthoma, and revealed that two of them showed a novel somatic missense mutation in exon6 of the 3-hydroxysteroid dehydrogenase (NSDHL) gene, and no mutation of exon4 was found in any case.[9] It is of interest that the lesion of our patient was on the thumb. Also she had neither evidence of any disease except verruciform xanthoma nor obvious history of any local infection or trauma in the thumb.
Therapeutic options for verruciform xanthoma included topical steroids, carbon dioxide laser, cryotherapy, wireloop electrosectioning, and radiation therapy. But none of these was completely effective and safe. Guo et al. had successfully treated multiple lesions on the vulva with imiquimod in a 2-year-old girl.[10] Anyways, the surgical excision is the most effective treatment for verruciform xanthoma to date. The lesions did not reappear after excision in our patient.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Shafer WG. Verruciform xanthoma. Oral Surg Oral Med Oral Pathol 1971;31:784-9.
[Google Scholar]
|
| 2. |
Bittar M, Happle R. CHILD syndrome avant la lettre. J Am Acad Dermatol 2004;50 2 Suppl: S34-7.
[Google Scholar]
|
| 3. |
Hegde U, Doddawad VG, Sreeshyla H, Patil R. Verruciform xanthoma: A view on the concepts of its etiopathogenesis. J Oral Maxillofac Pathol 2013;17:392-6.
[Google Scholar]
|
| 4. |
Philipsen HP, Reichart PA, Takata T, Ogawa I. Verruciform xanthoma – Biological profile of 282 oral lesions based on a literature survey with nine new cases from Japan. Oral Oncol 2003;39:325-36.
[Google Scholar]
|
| 5. |
Ide F, Obara K, Yamada H, Mishima K, Saito I, Kusama K, et al. Cellular basis of verruciform xanthoma: Immunohistochemical and ultrastructural characterization. Oral Dis 2008;14:150-7.
[Google Scholar]
|
| 6. |
Tang R, Kopp SA, Cobb C, Halpern AV. Disseminated verruciform xanthoma: A case report. Cutis 2014;93:307-10.
[Google Scholar]
|
| 7. |
Chyu J, Medenica M, Whitney DH. Verruciform xanthoma of the lower extremity – Report of a case and review of literature. J Am Acad Dermatol 1987;17:695-8.
[Google Scholar]
|
| 8. |
Mountcastle EA, Lupton GP. Verruciform xanthomas of the digits. J Am Acad Dermatol 1989;20:313-7.
[Google Scholar]
|
| 9. |
Mehra S, Li L, Fan CY, Smoller B, Morgan M, Somach S. A novel somatic mutation of the 3-beta-hydroxysteroid dehydrogenase gene in sporadic cutaneous verruciform xanthoma. Arch Dermatol 2005;141:1263-7.
[Google Scholar]
|
| 10. |
Guo Y, Dang Y, Toyohara JP, Geng S. Successful treatment of verruciform xanthoma with imiquimod. J Am Acad Dermatol 2013;69:e184-6.
[Google Scholar]
|
Fulltext Views
3,569
PDF downloads
1,842





