Translate this page into:
Zosteriform cutaneous metastasis of renal cell carcinoma
2 Department of Pathology, Gulhane School of Medicine, Ankara, Turkey
Correspondence Address:
Mustafa Tunca
GATA Dermatoloji AD. Etlik, Kecioren, Ankara 06018
Turkey
| How to cite this article: Dincer D, Tunca M, Akar A, Kurt B. Zosteriform cutaneous metastasis of renal cell carcinoma. Indian J Dermatol Venereol Leprol 2011;77:340-342 |
Sir,
A 45-year-old man presented to our outpatient clinic with numerous, quite firm, hyperpigmented to violaceous, well-defined papules mostly ranging from a pinpoint to a few millimeters, up to 0.5 cm in diameter, located on his anterior left lower chest in a zosteriform distribution which persisted for more than a month [[Figure - 1]a and b]. He had a history of stage IV renal cell carcinoma (RCC) involving the right kidney, diagnosed 7 years ago. His case records showed that he had cytoreductive right radical nephrectomy, right suprarenalectomy and wedge resection; for the primary tumor, metastatic adrenal and metastatic corpus sternum tumors, respectively. Meanwhile, he received three courses of radiotherapy and chemotherapy; he also got medical therapies including interferon and an angiogenesis/multikinase inhibitor, sunitinab. A month before he was admitted to our clinic, he had developed zosteriform papules over the lower part of the anterior chest wall on the left side which erupted suddenly within days, accompanied by mild pain and deep dermal tenderness. He had been diagnosed to have herpes zoster by a dermatologist. Antiviral treatment was initiated but the lesions had not responded.
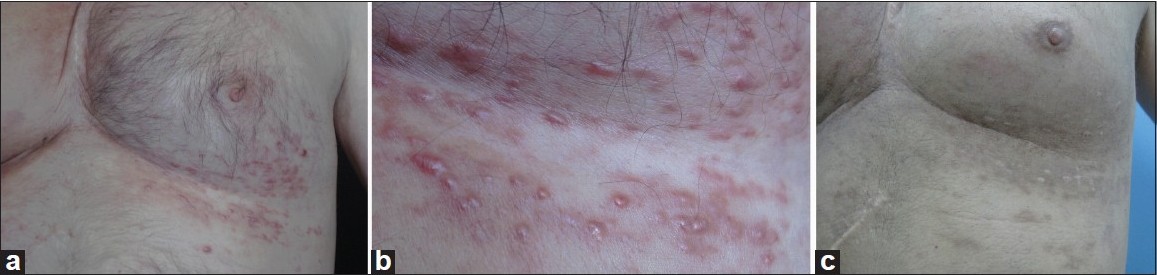 |
| Figure 1: (a) Zosteriform appearance of metastatic lesions over the lower anterolateral part of the chest on the left side. (b) Close-up view of the firm, hyperpigmented to violaceous, well-defined papules mostly ranging from pinpoint to a few millimeters in diameter. (c) Clinical picture of the patient after radiotherapy, 7 months later |
Upon admission to our clinic, one of these lesions was punch biopsied. A light microscopic evaluation of the hematoxylin and eosin (H and E)-stained histopathological slides showed a diffuse, solid, dense dermal infiltrate of atypical epithelial cells. Neoplastic cells had a pleomorphic nucleus, and a large nucleolus with pale cytoplasm [[Figure - 2]a]. These tumor cells were identical with patient′s previous renal cell carcinoma cells [[Figure - 2]b].
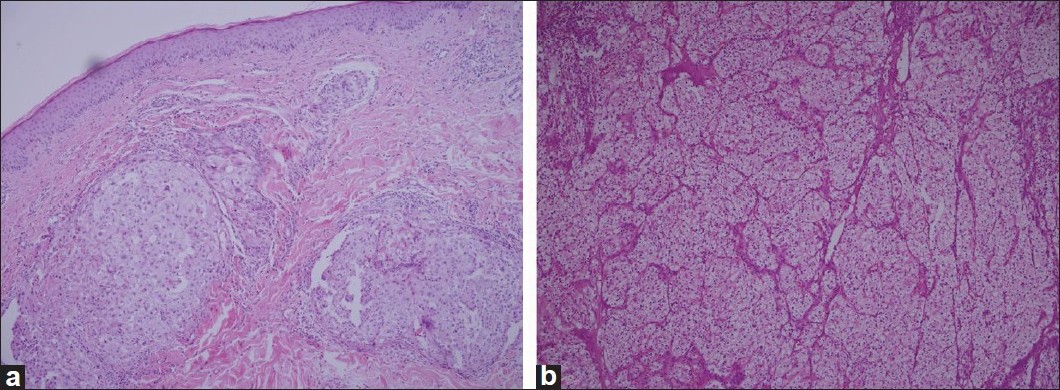 |
| Figure 2: (a) Histopathologic view of the metastatic renal cell carcinoma: diffuse, solid, dense dermal infiltrate of neoplastic epithelial cells with a pleomorphic nucleus and a large nucleolus with pale cytoplasm. (b)- These cells were identical with the tumor cells of renal cell carcinoma resected from kidney 7 years ago (both slides H and E stained, ×100) |
With a higher magnification, a high mitotic activity was noted. We also noted a peripheral nerve branch near the tumor mass [Figure - 3]. To verify diagnosis, both original and metastatic tumors were stained immunohistochemically with CD10. Both of them were positive and the patient was diagnosed as having metastatic RCC. In computed tomographic evaluation, findings of metastasis to left adrenal gland, mass formation at the right suprarenal region, and another mass formation with irregular borders in the anterior mediastinal region, adjacent to ascending aorta, were detected.
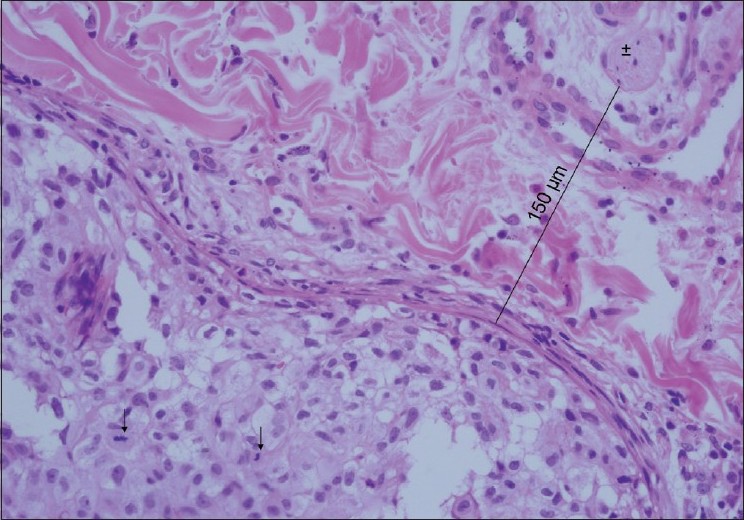 |
| Figure 3: With higher magnification, high mitotic activity can be seen. This figure also shows a nerve branch near the tumor mass (H and E, ×400), ↑ : Mitotic cells, ±: Nerve branch |
The skin is a relatively uncommon site for metastasis of internal malignancies and most common primary sites are breast, lung, gastrointestinal system, and oral mucosa. [1] The most common clinical presentation of cutaneous metastasis is nodule(s) or mass(es). In our case, both the primary tumor (RCC) and clinical presentation (zosteriform pattern) are rare features. [2],[3] When we searched PubMed for zosteriform cutaneous metastatic RCC, we only detected an abstract of a case which was presented as a poster in 2004. [4]
The exact mechanism of zosteriform metastases is not known. Several possible explanations have been proposed. One possible mechanism is increased susceptibility at the site of previous herpes zoster. Perineural lymphatic spread, and spread via fenestrated vessels of the dorsal root ganglion are other theories and in some cases accidental surgical implantation was blamed. [3],[4] Our patient did not have a history of previous herpes zoster or surgery in the metastatic tumor site. The most likely mechanism responsible for zosteriform distribution in our patient may be perineural lymphatic spread. In histopathological examination, we noted a nerve branch near the tumor mass. At one point, the distance from the tumor mass to a nerve branch was measured to be 150 ΅m [Figure - 3]. This may be a clue for perineural lymphatic spread and may also explain mild pain in our patient.
It has been shown that in most patients with cutaneous metastasis of internal malignancies, there is other organ or lymph node involvement at the time of diagnosis of cutaneous metastasis. [5] Therefore, cutaneous metastasis is a sign that the disease is already widespread and the prognosis is poor. In our case, we have searched for possible metastases and found evidence of metastatic lesions in both adrenal regions and in the anterior mediastinal region.
The management of RCC is dependent on the status of the patient and the number of metastatic lesions. The treatment for localized cutaneous metastatic lesions is usually surgical. Radiotherapy and/or chemotherapy have much of a role in extensively disseminated cases. Cutaneous lesions in our case resolved with radiotherapy [[Figure - 1]c]. However, several new lesions began developing on the right lateral chest wall 7 months later, and these lesions were also treated with radiotherapy.
In conclusion, this case shows that the clinical presentation of skin metastasis of RCC is variable, and although very rare, lesions may occur in a zosteriform pattern. Dermatologists should also remember that in most cases, cutaneous metastasis is a sign of widespread metastatic disease and warrants intensive search for other possible metastatic tumors elsewhere in the body.
| 1. |
Hu SC, Chen GS, Wu CS, Chai CY, Chen WT, Lan CC. Rates of cutaneous metastases from different internal malignancies: Experience from a Taiwanese medical center. J Am Acad Dermatol 2009;60:379-87.
[Google Scholar]
|
| 2. |
LeSueur BW, Abraham RJ, DiCaudo DJ, O'Connor WJ. Zosteriform skin metastases. Int J Dermatol 2004;43:126-8.
[Google Scholar]
|
| 3. |
Rao R, Balachandran C, Rao L. Zosteriform cutaneous metastases: A case report and brief review of literature. Indian J Dermatol Venereol Leprol 2010;76:447.
[Google Scholar]
|
| 4. |
Davidson KA, Wong H. Zosteriform metastatic renal cell carcinoma. (Poster Abstract) J Am Acad Dermatol 2004;50:P113.
[Google Scholar]
|
| 5. |
Saeed S, Keehn CA, Morgan MB. Cutaneous metastasis: A clinical, pathological, and immunohistochemical appraisal. J Cutan Pathol 2004;31:419-30.
[Google Scholar]
|
Fulltext Views
1,565
PDF downloads
1,231





