Translate this page into:
Coxsackievirus B5–induced severe mucocutaneous reaction mimicking drug-induced Stevens–Johnson syndrome/toxic epidermal necrolysis
2 School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan
3 Department of Pediatrics, Wan Fang Hospital, Taipei Medical University, Taipei, Taiwan
4 Department of Dermatology, Wan Fang Hospital, Taipei Medical University; School of Medicine, College of Medicine, Taipei Medical University, Taipei, Taiwan
Correspondence Address:
Yu-Chen Huang
111, Hsing-Long Road Sec. 3, Wenshan District, Taipei City 116
Taiwan
| How to cite this article: Tsai TY, Chao YC, Lai YH, Huang YC. Coxsackievirus B5–induced severe mucocutaneous reaction mimicking drug-induced Stevens–Johnson syndrome/toxic epidermal necrolysis. Indian J Dermatol Venereol Leprol 2019;85:416-418 |
Sir,
Group B coxsackieviruses cause hand-foot-and-mouth disease and herpangina. Recently, enterovirus and Coxsackie A6 virus induced severe mucocutaneous reactions resembling drug-induced Stevens–Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN), both clinically and histopathologically, have been reported.[1],[2] Here, we report the first case of coxsackievirus B5–induced severe mucocutaneous reaction mimicking drug-induced SJS/TEN.
A 12-year-old apparently healthy boy presented to our emergency department with a 2-day history of generalized skin rashes. It was preceeded by fever and rhinorrhea since last 6 days. Congested/painful eyes and multiple buccal ulcers developed subsequently. He was initially diagnosed with herpangina. However, maculopapular rashes developed on his trunk, with rapid progression to extremities and face. He reported past treatment with ibuprofen, acetaminophen, dextromethorphan, diclofenac, cephalexin and sulfamethoxazole eye drops, without any appreciable benefit. Initially, ibuprofen, diclofenac, and cephalexin were suspected to be the culprit agents for suspected drug eruptions. However, absence of similar symptoms during previous treatment with these drugs refuted our suspicion.
Physical examination revealed widespread, dusky-red, purpuric, maculopapular rashes with vesicles on the face, trunk, extremities, palms, and soles [Figure - 1]; conjunctival hyperemia with purulent discharge resulting in eyelid adhesion [Figure - 2]a; ulcers on his buccal mucosa and hard palate; and crusted hemorrhagic erosions on the lips [Figure - 2]b, scrotum, and glans penis. Hair and nails were spared. Initial differential diagnoses included drug-induced SJS/TEN, Mycoplasma pneumoniae–induced rash and mucositis, erythema multiforme major, and atypical hand-foot-and-mouth disease.
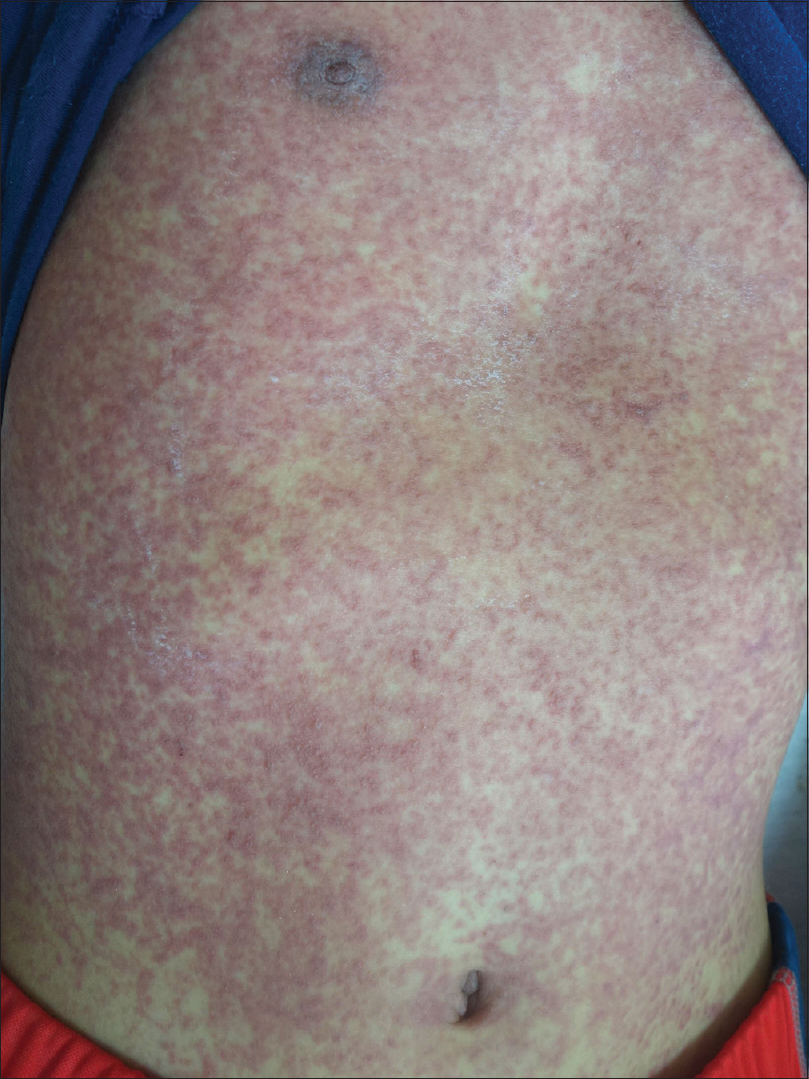 |
| Figure 1: Widespread, dusky-red, purpuric maculopapular rashes with confluence and vesicles on the trunk |
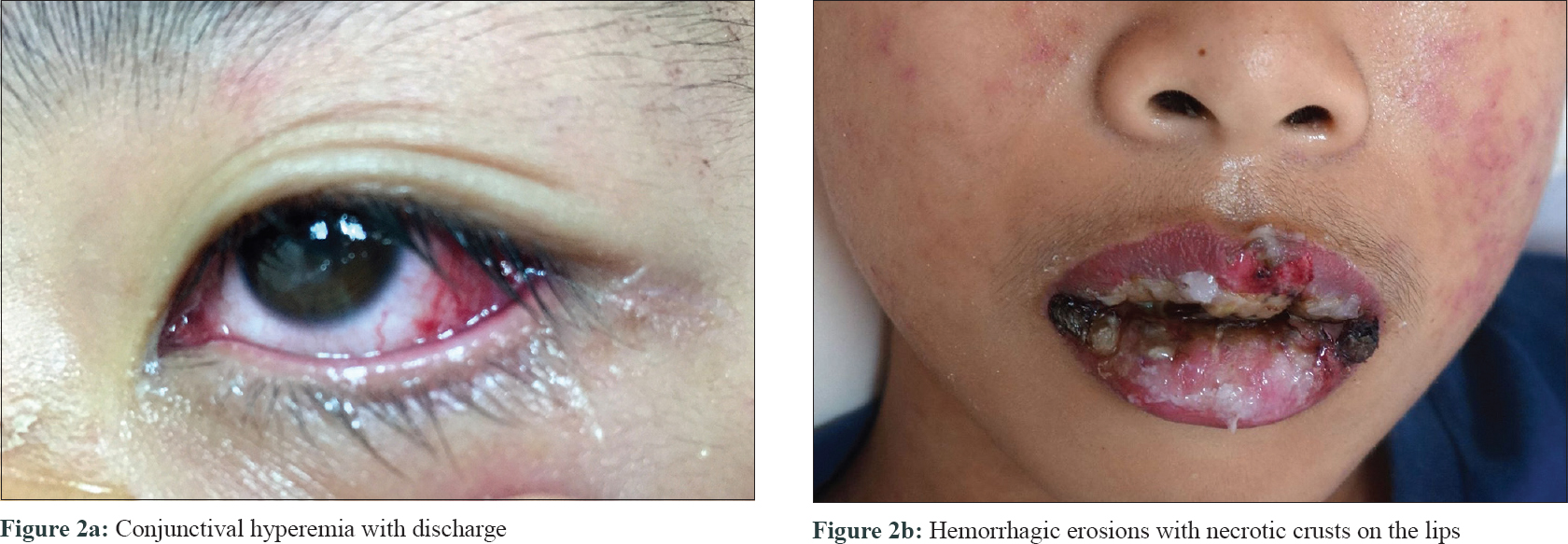 |
| Figure 2: |
Laboratory investigations revealed elevated c-reactive protein level (22.4 mg/dL) without leukocytosis, anemia, thrombocytopenia, or abnormal creatinine level. Types 1 and 2 herpes simplex virus IgG and IgM were both negative. Mycoplasma pneumoniae antibody and IgM analysis were equivocal.
We administered cefazolin and methylprednisolone after admission resulting in dusky discolouration of skin and gradual desquamation. Notably, ciliary loss, severe purulent conjunctivitis, synechiae due to pseudomembrane formation and corneal scarring were observed. Prednisolone, levofloxacin, and cyclosporine eye drops were administered, along with mechanical removal of the pseudomembrane.
Skin biopsy demonstrated subepidermal blisters, vacuolar degeneration of the basal layer, scattered necrotic and apoptotic keratinocytes, and perivascular lymphohistiocytic and eosinophilic infiltration in the dermis. These findings are consistent with both enterovirus-induced severe mucocutaneous reactions and drug-induced SJS/TEN. However, neutrophil-rich intraepidermal infiltration in our case narrowed the diagnosis to enterovirus-induced severe mucocutaneous reactions.[2],[3]
Blood, wound, and throat swab cultures were unremarkable. The patient recovered well and was discharged after 20 days. One week after discharge, the patient presented to us with mild cutaneous hyperpigmentation and onychomadesis involving all the 10 nails. One month later, the patient was diagnosed with coxsackievirus B5–induced severe mucocutaneous reaction, corroborated by the presence of coxsackievirus B5 in throat swab viral culture. Meanwhile, a lymphocyte transformation test measuring T-cell proliferation against drugsin vitro was performed. The results were negative for acetaminophen, diclofenac, ibuprofen, cephalexin, and trimethoprim–sulfamethoxazole, which greatly reduced the probability of drug-induced SJS/TEN. However, lymphocyte transformation test has limited sensitivity of 60%–70% and specificity of at least 85%.[4]
Our case demonstrates the etiological role of coxsackievirus B5 in severe mucocutaneous reactions resembling drug-induced SJS/TEN. Notably, mucosal involvement is a cardinal symptom of drug-induced SJS/TEN. In contrast, ocular and genital mucous involvement is rarely observed in enterovirus infection. Moreover, in severe drug-induced SJS/TEN cases, ocular pseudomembrane formation and corneal scarring may occur, as observed in the present case.[3] Interestingly, similar severe ocular involvement has not been observed in coxsackievirus A6–induced severe mucocutaneous reactions.[1],[2]
Cutaneous manifestations of enterovirus-induced severe mucocutaneous reactions and drug-induced SJS/TEN may range from maculopapules and vesicles to erosions. However, drug-induced SJS/TEN is characterized by target-like lesions, which were absent in the present case.[1],[2],[3] Complex interactions between viral infections and drug allergy have been observed in some clinical senarios. For example, extensive maculopapular rashes develop in patients with infectious mononucleosis after treatment with ampicillin. Nervertheless, similar viral–drug interaction has not yet been reported in coxsackievirus infection.[5]
To conclude, we report an unusual case illustrating the potential of coxsackievirus B5 to induce clinical symptoms that closely resemble drug-induced SJS/TEN. Thus, we should maintain a high index of suspicion for enterovirus infection while evaluating patients with severe mucocutaneous symptoms.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient has given his consent for his images and other clinical information to be reported in the journal. The patient understand that name and initials will not be published and due efforts will be made to conceal identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Huang WC, Huang LM, Lu CY, Cheng AL, Chang LY. Atypical hand-foot-mouth disease in children: A hospital-based prospective cohort study. Virol J 2013;10:209.
[Google Scholar]
|
| 2. |
Laga AC, Shroba SM, Hanna J. Atypical hand, foot and mouth disease in adults associated with coxsackievirus A6: A clinico-pathologic study. J Cutan Pathol 2016;43:940-5.
[Google Scholar]
|
| 3. |
Valeyrie-Allanore L, Roujeau JC. Epidermal necrolysis (Stevens-Johnson Syndrome and toxic epidermal necrolysis). In: Goldsmith LA, Katz SI, Gilchrest BA, Paller AS, Leffell DJ, Wolff K, editors. Fitzpatrick's Dermatology in General Medicine. Vol. 8. Ch. 40. New York: The McGraw-Hill Companies; 2012.
[Google Scholar]
|
| 4. |
Pichler WJ, Tilch J. The lymphocyte transformation test in the diagnosis of drug hypersensitivity. Allergy 2004;59:809-20.
[Google Scholar]
|
| 5. |
Levy M. Role of viral infections in the induction of adverse drug reactions. Drug Saf 1997;16:1-8.
[Google Scholar]
|
Fulltext Views
4,195
PDF downloads
905





