Translate this page into:
Is dermoscopy a useful tool in pseudo-Kaposi sarcoma?
Corresponding author: Dr. Inigo Navarro-Fernandez, Avenida Valdecilla, 25, 39008, Santander, Spain. inavarrofez@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Navarro-Fernandez I, Duran-Vian C, Gonzalez-Vela MC, Yange-Zambrano G, Gonzalez-Lopez MA. Is dermoscopy a useful tool in pseudo-Kaposi sarcoma? Indian J Dermatol Venereol Leprol 2021;87:709-2.
Sir,
Pseudo-Kaposi sarcoma, also known as acroangiodermatitis, is a disorder characterized by the presence of a reactive proliferation of blood vessels that usually takes place on the patients’ limbs in the setting of different vascular disruptions. These include chronic venous insufficiency in acroangiodermatitis of Mali and congenital arteriovenous malformations in Stewart-Bluefarb syndrome.1,2
Clinically, patients present with violaceous papules that can coalesce into plaques, mimicking Kaposi sarcoma [Figure 1a]. On histological examination, pseudo-Kaposi is characterized by lobular vascular proliferation with associated perivascular fibrosis. Erythrocyte extravasation and dermal siderophages are frequently encountered [Figure 1b].
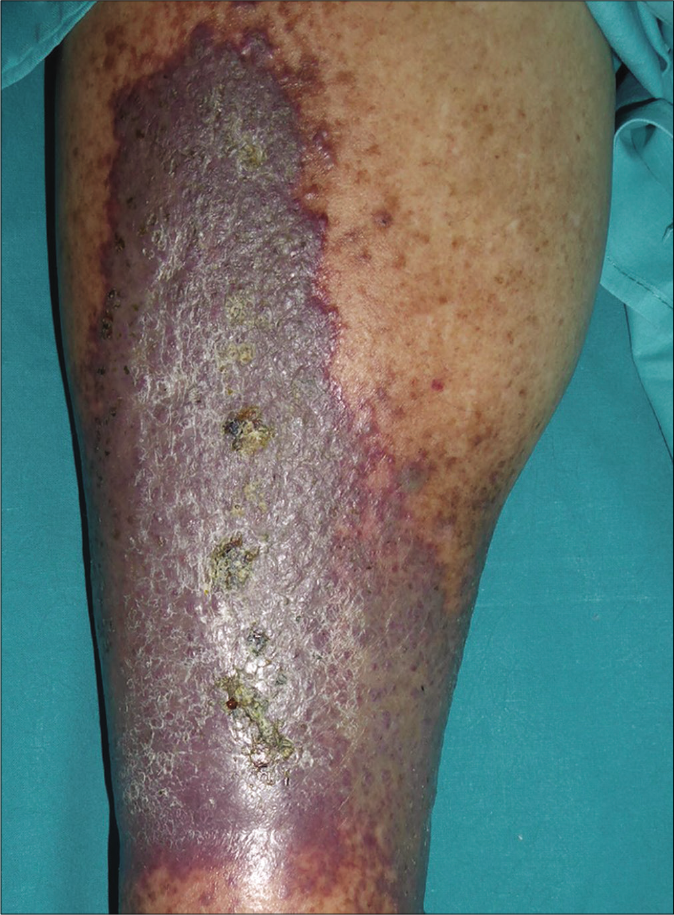
- Clinical photograph of one of the patients’ lower leg showing the classical features of acroangiodermatitis
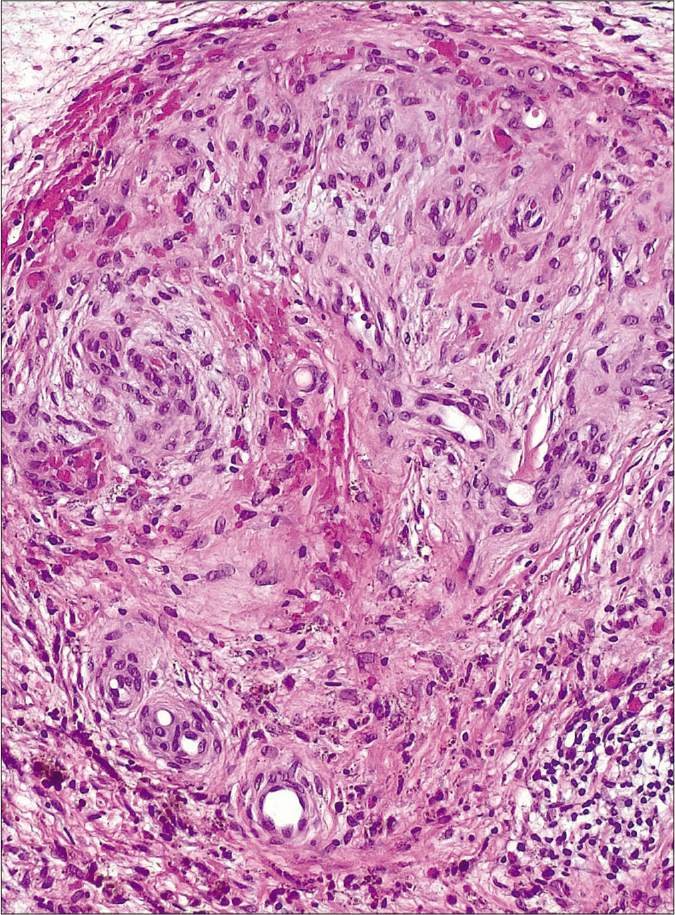
- Histopathological image of one of the patients showing an exuberant proliferation of vessels with a lobular configuration. Red blood cell extravasation and hemosiderin deposition can be observed (H and E, ×200)
In this report, we describe the dermoscopic features of four cases of pseudo-Kaposi. We found only three previous dermoscopic descriptions in the literature. All patients were treatment naïve when they were first seen in our clinic, except for patient two, who had been using a furoate mometasone ointment for the past month with partial relief of his symptoms. In every case, the diagnosis was suspected clinically and confirmed with histopathological and immunohistochemical examination: CD34 was expressed in endothelial cells but not in the surrounding perivascular cells, whereas immunostaining was negative for human herpesvirus 8 (HHV-8) in all cases.
Clinical signs of chronic venous insufficiency were obvious in patients one, two and four, whereas patient three had suffered a previous deep venous thrombosis (diagnosed by Doppler ultrasound) three weeks before the lesions started to develop.
In preceding reports, irregular vessels and white structureless areas were described in two cases,2 while white rail lines, red and blue lacunae and hemorrhagic crusts were found on the other one.3 We believe that, overall, different terms were used to refer to similar structures.
The clinical and dermoscopic features of our patients are outlined in Table 1. In all cases, a clearly vascular pattern was appreciated. All of them presented a mauve to violaceous background color with polymorphic vessels which reflect the vascular proliferation (the red lacunae probably embodying the lobular proliferation) [Figure 2]. Polychromatic color change (or rainbow pattern), like the one described in Kaposi sarcoma, was demonstrated in three patients. While this was initially described as a specific sign for Kaposi sarcoma, it has now also been described in many other lesions, such as angiokeratomas, hypertrophic scars, melanoma and dermatofibroma.4 The exact mechanism by which this phenomenon occurs is still not well established. However, it is probably related to the different refraction indices of polarized light which exist throughout a disorganized dermis with increased vascularity.4
| Patient | Sex | Age | Duration of the disease | Likely cause | Clinical examination | Dermoscopic features |
|---|---|---|---|---|---|---|
| 1 | Female | 92 | 2 years | CVI | Bilateral violaceous plaques with superficial crusting, on the lower legs. | Bluish background, polymorphic vessels, rainbow pattern, hemorrhagic crusts, WSS, sticky fiber sign |
| 2 | Male | 77 | 6 months | CVI | Bilateral vascular looking plaques with satellite nodules and superficial scale, distally on the lower limbs. | Polymorphic vessels, vascular lacunae, rainbow pattern, hemorrhagic crusts, WSS, peripheral coppery dots |
| 3 | Male | 57 | 4 months | DVT | Unilateral shiny violaceous papules coalescing into a large plaque on the right leg | Polymorphic vessels, vascular lacunae, rainbow pattern, WSS |
| 4 | Male | 87 | 9 months | CVI | Bilateral shiny violaceous papules and plaques with intense scaling on the distal lower extremities | Polymorphic vessels, vascular lacunae, WSS |
CVI: Chronic venous insufficiency, DVT: Deep venous thrombosis, WSS: White shiny structure
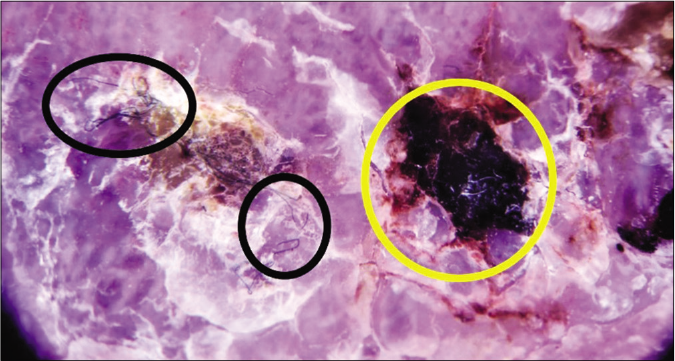
- Dermlite DL200 HR with polarized light (×10). Dermoscopic image of patient 1 revealing hemorrhagic crusts (yellow circle) and the “sticky fiber” sign (black circles)
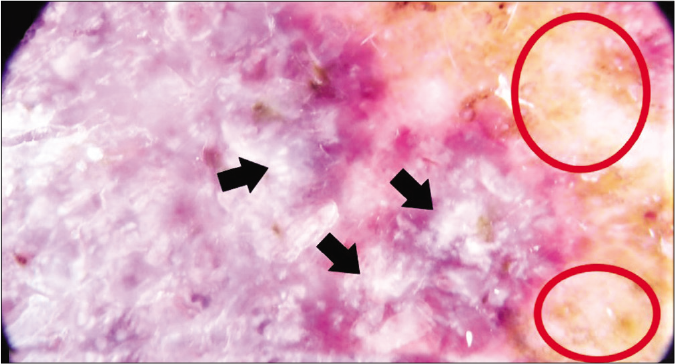
- Dermlite DL200 HR with polarized light (×10). Dermoscopic image of patient 2. Peripheral coppery dots (red circles) as well as white shiny structures (black arrows) are visible
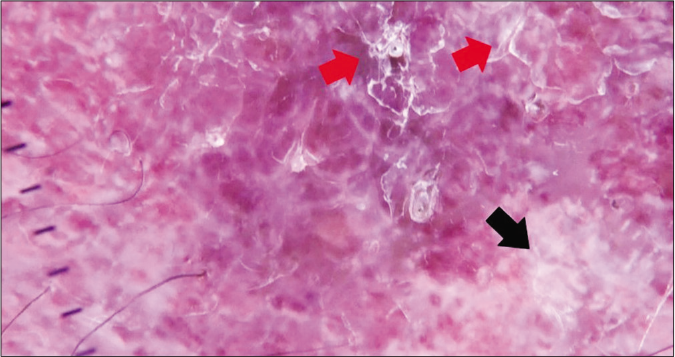
- Dermlite DL200 HR with polarized light (×10). Dermoscopic image of patient 3. White shiny structures (black arrows) and superficial scales (red arrows) can be observed
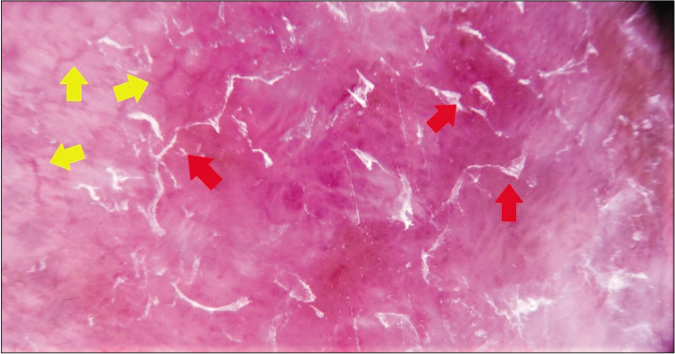
- Dermlite DL200 HR with polarized light (×10). Dermoscopic image of patient 4. Irregular vessels (yellow arrows) and superficial scales (red arrows) are pointed out
White shiny structures, encompassing white shiny lines, white shiny areas and rosettes, were seen in all cases. They may correlate with fibroblastic proliferation and are probably linked to the interaction of polarized light with collagen due to a birefringence phenomenon.5 Superficial white scales were also present in all cases. They are likely to reflect the orthokeratotic hyperkeratosis which is sometimes demonstrated on biopsies.
The “sticky fiber” sign was present in the first case. This clue reflects the presence of spongiosis, transudation, excoriation or ulceration in different lesions and we believe it highlights the transudation which takes place as a result of the increase in blood pressure in the lower legs.6 In the second case, a peripheral, coppery pigmentation was also visible. This is likely to be secondary to the associated stasis dermatitis which causes extravasation of red blood cells and hemosiderin deposits. Thus, in these cases, dermoscopy was not only useful in establishing the diagnosis of this syndrome, but it also contributed to a better understanding of its etiology and its link to venous insufficiency.
The pathogenesis of acroangiodermatitis is still poorly understood. However, it has been suggested that it might potentially develop as a consequence of increased venous and capillary pressure leading to an angiogenic response. This response could be mediated by different angiogenic factors, including vascular endothelial growth factor and PGE1 which stimulate the proliferation of fibroblasts and endothelial cells.7 This blood pressure increase is also presumably responsible for the extravasation of hemosiderin that can be observed both dermoscopically and histologically.
Management of acroangiodermatitis should be focused on treating the underlying cause (including limb elevation and compressive bandages in cases secondary to chronic venous insufficiency and vascular surgery for arterovenous malformation).
When analyzing the morphological patterns of these cases (as well as the ones previously described in the literature), there seem to be certain structures which are present most of the time. These include vascular lacunae, polymorphic vessels, white shiny structures and even polychromatic color change.
We, therefore, believe that dermoscopy can be useful in distinguishing acroangiodermatitis from other conditions which can mimic it clinically. These include lichen planus (violet hue without irregular vessels), angiokeratomas (presence of dark lacunae without polymorphic vessels), pigmented purpuric dermatitis (absence of white shiny structures), lichen aureus (lack of white shiny structures, less prominent vascular pattern) and lichen simplex chronicus (faint vascularity with no lacunae). For differentiating from Kaposi sarcoma, there are some dermoscopic clues which could be explored. For instance, increased fibroblastic activity in pseudo-Kaposi sarcoma presenting as white shiny structures might be suggestive of this diagnosis. However, differentiation of both disorders based solely on clinical and dermoscopic grounds is virtually impossible in some cases. Consequently, we believe histopathology with immunostaining for HHV8, D2-40 and CD34 that remains the gold standard for the diagnosis of this disease.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Acroangiodermatitis of mali: A rare vascular phenomenon. Indian J Dermatol Venereol Leprol. 2010;76:553-6.
- [CrossRef] [Google Scholar]
- Dermoscopic features in two cases of acroangiodermatitis. Australas J Dermatol. 2018;59:e290-1.
- [CrossRef] [Google Scholar]
- Pseudo-Kaposi sarcoma: Report of a case investigated by dermoscopy, reflectance confocal microscopy and optical coherence tomography. J Eur Acad Dermatol Venereol. 2018;32:e429-32.
- [CrossRef] [Google Scholar]
- The rainbow pattern in dermoscopy: A zoom on nonkaposi sarcoma skin diseases. Biomed J. 2018;41:209-10.
- [CrossRef] [Google Scholar]
- Observation of chrysalis structures with polarized dermoscopy. Arch Dermatol. 2009;145:618.
- [CrossRef] [Google Scholar]
- Pseudo-Kaposi's sarcoma (Bluefarb-Stewart type) Int J Dermatol. 2005;44:136-8.
- [CrossRef] [Google Scholar]





