Translate this page into:
Renal function status in leprosy
Correspondence Address:
P K Nigam
Katora Talab, Raipur - 492 001, M.P.
India
| How to cite this article: Singh S, Nigam P K, Sethi I M. Renal function status in leprosy. Indian J Dermatol Venereol Leprol 1997;63:162-165 |
Abstract
75 leprosy patients comprising of 47 males and 28 females ranging in age from 10 years to 75 years (mean 33.76 years) were studied. Duration of disease ranged from one month to 12 years. Thirty (40%) cases were in reaction. Renal involvement occurred in 53.66% of cases. 80% of type 2 reaction leprosy patients showed renal involvement as compared to 53% of type 1 reaction. 80% of leprosy cases with more than 3 years duration of disease showed renal involvement as compared to 46.6% of cases with less than 3 years duration of disease. 9% of cases showed reduction in one or both kidney size with loss of corticomedullary differentiation on USG.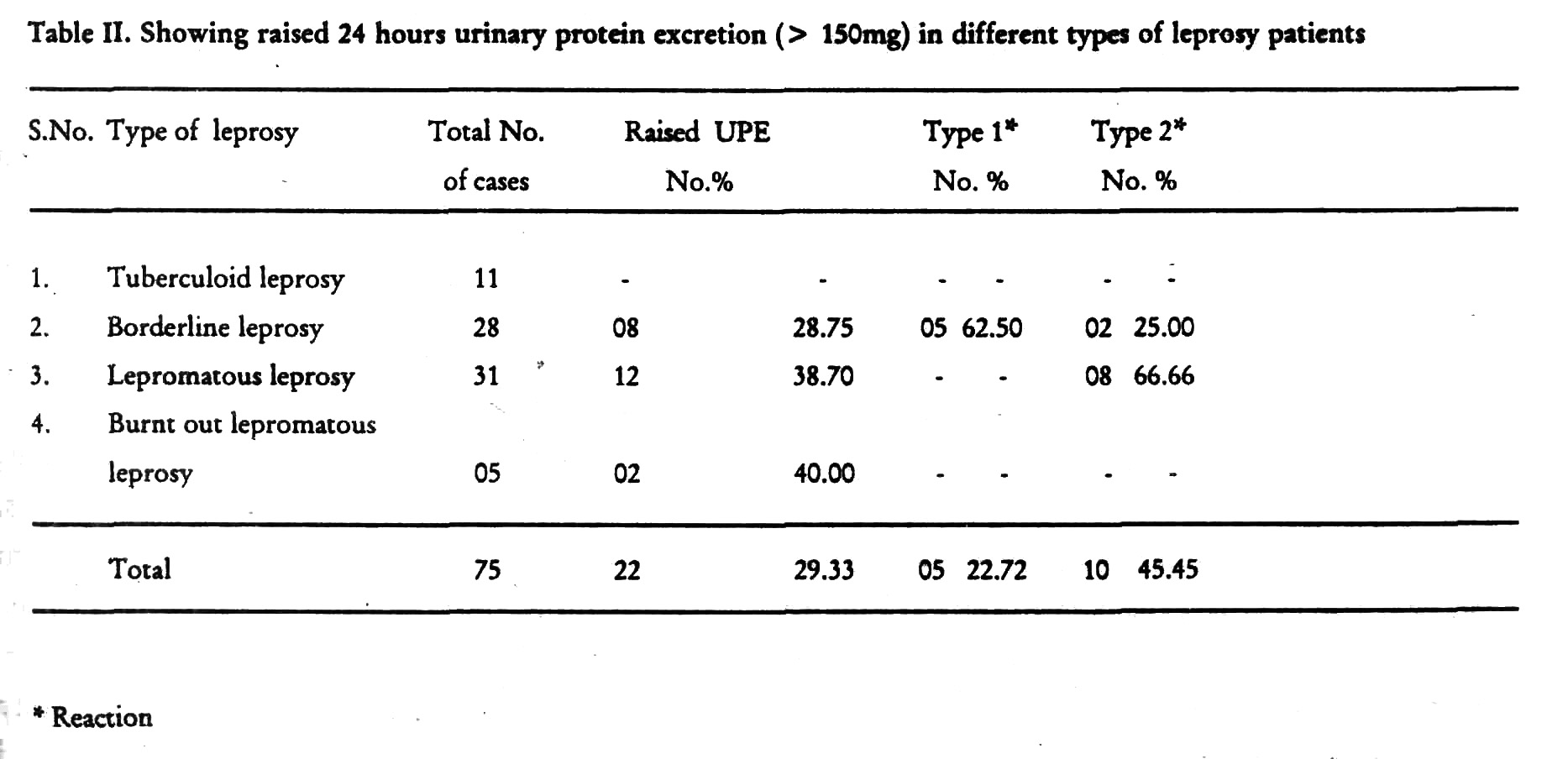


 |
 |
Although M leprae ordinarily does not invade the renal parenchyma, considerable renal function impairment in leprosy has been reported.[1][2] However, most patients with renal involvement have only asymptomatic urinary abnormalities. Renal biopsy in leprosy patients has shown variety of lesions including (1) secondary amyloidosis, (2) glomerulonephritis, (3) distal tubular function defects, (4) pyelonephritis, (5) interstitial nephritis, and patients may present with nephrotic syndrome-like picture. Renal involvement in leprosy may progress to end stage renal disease and may be an important cause of death in leprosy patients.[3]
The present study was undertaken to evaluate the renal functional impairment in leprosy patients using clinical, biochemical and ultrasonographical criteria.
Subjects and Methods
Seventy-five patients of leprosy were studied. The patients were thoroughly examined and a detailed history regarding the disease, its duration, treatment and reaction in the past were recorded. Those patients who had associated history of renal disease, diabetes mellitus and hypertension were excluded from the study. Slit-skin smear examination was carried out in all the cases. Routine investigations including total and differential leucocyte count, Hb%, ESR, blood sugar, blood urea and serum cholesterol were done. Serum creatinine estimation was done by modified Jaffe′s method. Total serum protein, albumin and globulin were estimated by Biuret and BCG binding method. Ultrasonographic study (USG) of kidneys was done in all the cases using 3.5 MCH real time sector scanner.
Results
Out of 75 patients, 47 (62.66) were males and 28 (37.45%) females. The age of the patients ranged from 10 years to 75 years (mean age 33.76 years). The duration of disease ranged from one month to 12 years. Thirty of the patients were in reaction. Of these, 15 were in type 1 reaction (2 belonged to borderline and 13 patients to lepromatous leprosy).
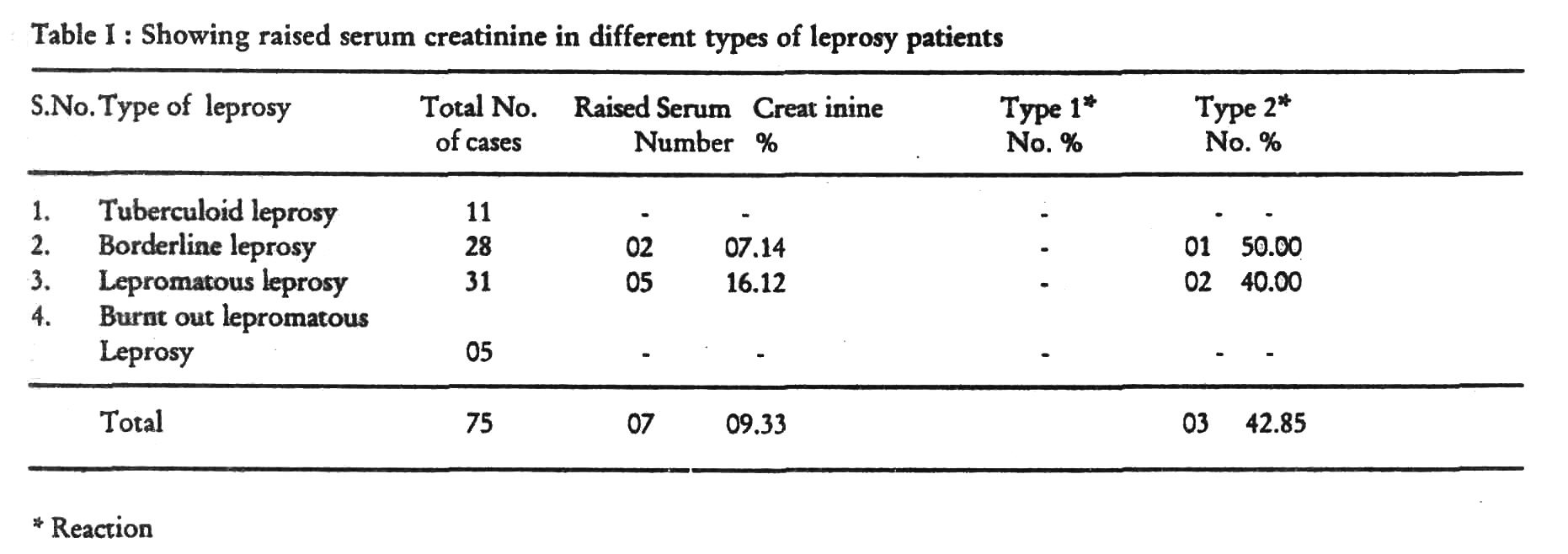
The renal involvement was observed in all the age groups and both the sexes, equally and it was found in 40 (53.66%) of cases, 9% of the tuberculoid patients, 50% of the borderline leprosy, 70% of the lepromatous leprosy cases and 60% of the burnt-out lepromatous leprosy cases. The incidence of renal involvement increased with the duration of leprosy. Leprosy patients with reaction had significantly higher incidence of renal involvement than those without reaction (P<.01). Further, 80% of leprosy patients with type 2 reaction showed renal involvement as compared to 53.3% of type 1 reaction.
Nine of the 75 patients had haematuria. Out of these three patients were in type 1 reaction and four were in type 2 reaction. Proteinuria was observed in 10.66% cases. Out of those, 6 (19.35%) cases were of lepromaous leprosy in which 4(66.66%) were in type 2 reaction. One patient each of borderline and burnt out lepromatous leprosy also had proteinuria. Epithelial cell casts were present in 7 (9.33) of the 75 leprosy patients. Serum cholesterol values and the total serum protein values were within the normal range in all the patients. However, 28 patients had altered albumin globulin ratio.
Seven (9.33) patients had serum creati-nine value of more then 1.2 mg. [Table - 1]. Of these, 5 patients were of lepromatous leprosy case and one borderline in type 2 reaction.
Twenty-two (29.33%) of the 75 patients had increased 24 hours urinary protein excretion (UPE) of more than 150 mg. [Table - 2]. Eight (66.66%) of the 12 lepromatous leprosy patients with raised 24 hours UPE were in type 2 reaction. 31 (41.33%) of the 75 patients showed decreased creatinine clearance. The glomerular filtration rate (GFR) was also
found to be decreased in 31 of the 75 patients. None of the patients of tuberculoid leprosy, with or without reaction, had abnormal 24 hours UPE, creatinine clearance rate or GFR.
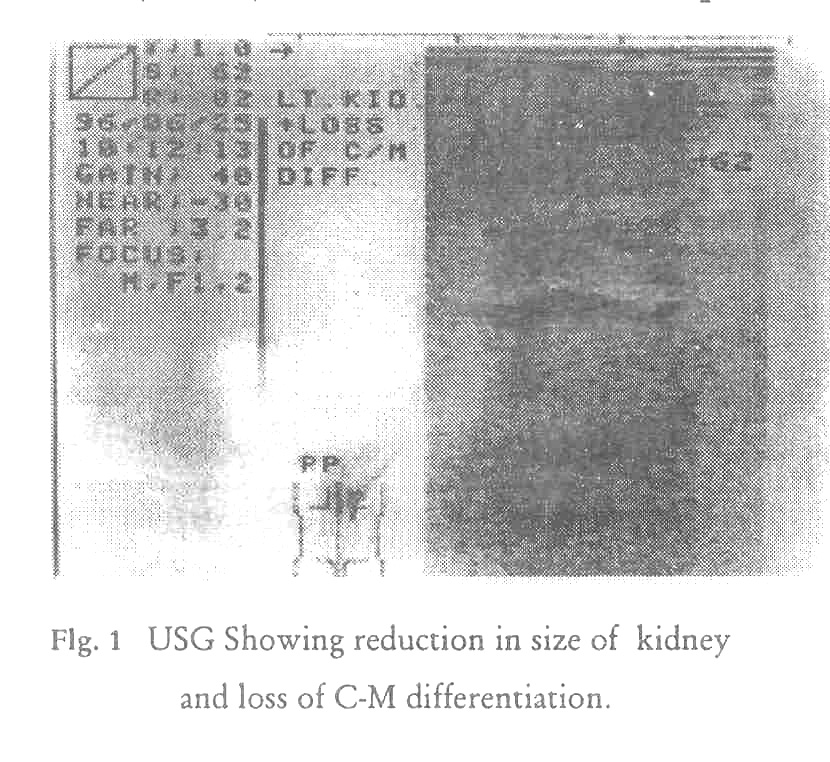
Seven (9.45%) patients (5 lepromatous leprosy and one each of borderline and burnt out lepromatous leprosy) showed some renal abnormality on USG Study. Out of these patients, 5 patients had bilateral reduction of renal size. Corticomedullary differentiation (CMD) was lost in 3 of these patients [Figure - 1]. One patient had unilateral reduction in renal size with loss of CMD. In one case of lepromatous leprosy, only the loss of bilateral CMD was found.
Discussion
Considerable renal function impairment, mostly due to circulating immune complexes, is known to occur in leprosy. Raised serum creatinine, raised 24 hours UPE, [5,6] and decreased GFR,[6] have been observed in leprosy patients. Reduced kidney size on USG study is an early sign of chronic renal failure,[7] and it was observed in 7(9.5%) of our patients. Only 9% of our tuberculoid leprosy patients had renal involvement as compared to 70% of lepromatous leprosy patients. This may be because these patients have maximum and recurrent chances of reaction which can cause deposition of immune complexes in the glomeruli, blood vessels and mesangium and lead to glomerulonephritis.[8]
| 1. |
Bedi TR, Kaur S, Singhal PC et al. Fatal proliferative glomerulonephritis in lepromatous leprosy, Lepr India, 1997;49:500-503.
[Google Scholar]
|
| 2. |
Kanwar AJ, Bharija SC, Belhaj MS. Renal function status in leprosy, Ind J Lepr, 1984;56:595-599.
[Google Scholar]
|
| 3. |
Bernard JC, Vasquez CAJ. Visceral lesion in lepromatous leprosy: study of 60 necropsies, Int J lepr, 1973;41:94-101.
[Google Scholar]
|
| 4. |
Date A, Thomas A, Mathai R et al. Glom-erular pathology in leprosy: an electron microscopic study, Am J Trop Med Hyg, 1997;26:256-272.
[Google Scholar]
|
| 5. |
Chopra NK, Lakhani JD, Pande RS et al. Renal involvement in leprosy, JAssoc Physicians India 1991;39:165-167.
[Google Scholar]
|
| 6. |
Nadeem M, Gar BR, Das AK. Renal functional status in leprosy, Ind J Dermatol Venereol Leprol 1995;61:133-136.
[Google Scholar]
|
| 7. |
Lcoe F, Brenner BM. Disorder of the kidney and urinary tract. In: Harrison's Parinciples of Internal Medicine, Vol-II, 13th edn, McGRow-HILL, Inc. 1994, p-1253.
[Google Scholar]
|
| 8. |
Glassock RJ, etal. Secondary glomerular disease, In : The Kidney, 4th edn. Editors, Brenner BM, Rector Jr, FC Saunder, Philadelphia, 1991, p-1280-1369.
[Google Scholar]
|





