Translate this page into:
Severe recalcitrant psoriasis treated with itolizumab, a novel anti-CD6 monoclonal antibody
Correspondence Address:
Aayush Gupta
B-402, The Metropolitan, Near Darshan Hall, Chinchwad, Pune - 411 033, Maharashtra
India
| How to cite this article: Gupta A, Sharma YK, Deo K, Kothari P. Severe recalcitrant psoriasis treated with itolizumab, a novel anti-CD6 monoclonal antibody. Indian J Dermatol Venereol Leprol 2016;82:459-461 |
Sir,
Psoriasis is an inflammatory autoimmune disease of varying severity. Severe psoriasis occurs in 20–30% of cases and is defined by body surface area involvement >10% and/or psoriasis area severity index >10 along with dermatology life quality index >10.[1] In such patients, the currently available systemic therapies may lack consistent efficacy over time, are potentially inconvenient and have the risk of serious cumulative toxicity.[2]
Improved understanding of the immunological basis of the pathophysiology of psoriasis has catalyzed the development of drugs targeting key pathogenetic steps; the ones used currently target tumor necrosis factor-α (infliximab, adalimumab and etanercept) or the Th17 pathway (ustekinumab).[3]
Itolizumab (Alzumab®, Biocon) is a novel humanized monoclonal antibody developed in India for moderate/severe chronic plaque psoriasis. It inhibits T-cell proliferation induced by activated leukocyte cell adhesion molecules and interleukin-2 by binding to the extracellular scavenger receptor cysteine rich distal domain 1 of CD6. It downregulates the phosphorylation of intracellular proteins and also reduces the production of interferon-γ, interleukin-6 and tumor necrosis factor-α.[2] We report its effect in a case of severe recalcitrant psoriasis.
A 22-year-old man presented to us in February 2014 with a history of persistent, nearly unrelenting psoriasis for 6 years. He had switched between many practitioners and between various therapeutic “disciplines” [Table - 1]. His general, physical and systemic examinations were normal. His weight was 50 kg. Dermatological examination revealed unstable severe psoriasis [Figure - 1]: psoriasis area severity index (PASI) score, 33.5; physician global assessment score, 5; nail psoriasis severity index score, 16 and dermatologic life quality index score, 18. Hematological, serological, radiological and biochemical parameters were normal. His Mantoux test was negative.

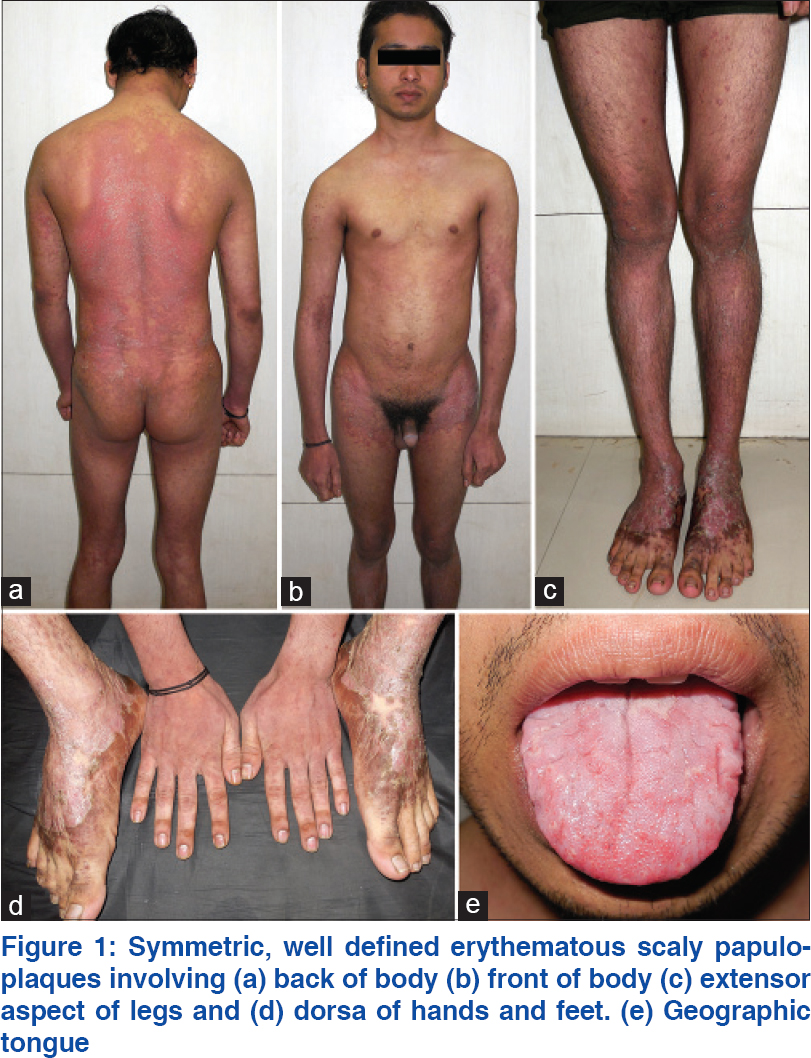 |
| Figure 1: Symmetric, well defined erythematous scaly papuloplaques involving (a) back of body (b) front of body (c) extensor aspect of legs and (d) dorsa of hands and feet. (e) Geographic tongue |
He was given cyclosporine, 200 mg daily for the first 2 weeks and infused with itolizumab, 75 mg (1.6 mg/kg in 250 ml of normal saline over 3 hours) fortnightly without any premedication. The PASI score dropped to 12.1 after 6 weeks and to 1.4 at the end of the induction phase after 12 weeks. There were only a few residual plaques persisting around his ankles at the end of 12 weeks [Figure - 2]. After the maintenance phase of three 4-weekly infusions, his PASI score was 1.8; physician global assessment score, nail psoriasis severity score and dermatologic life quality index score paralleled this improvement. There were no infusion reactions or other adverse effects. The patient was advised regular follow up and narrow band ultraviolet B biweekly, later replaced with topical corticosteroids following his inability to comply.
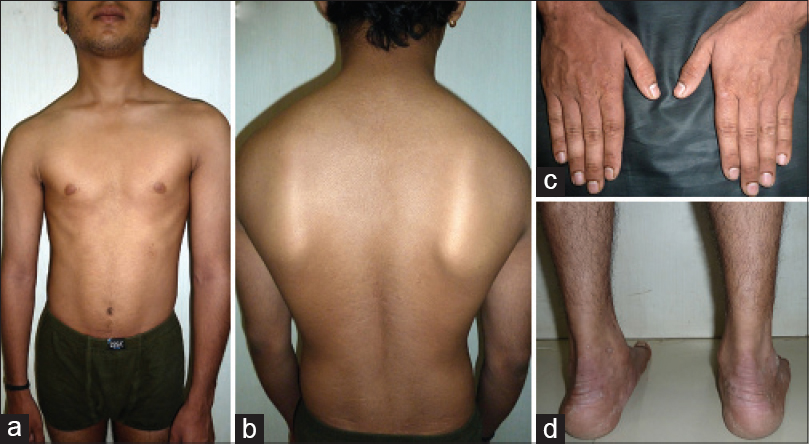 |
| Figure 2: At the end of 12 weeks, clearance over (a) front of the body (b) back of the body (c) dorsa of the hands. (d) Persistent lesions around the ankles |
Follow up after 15 months revealed that the PASI score had risen to 4, with some fresh papulo-plaques developing on the scalp in addition to the persistent lesions around the ankles. He has been counseled regarding the possible necessity of continuing infusions of itolizumab, in case of a severe relapse.
Cyclosporine, infliximab, acitretin, methotrexate and ustekinumab are considered first-line systemic therapies for control of severe psoriasis.[3] Concerns regarding the immunosuppressive effects and the increased risk of infections following tumor necrosis factor-α inhibitors in severe psoriasis clearly indicate the need for further search for alternative therapies which would inhibit T-cell activation rather than block its consequences.
Itolizumab, one such agent, is a humanized monoclonal antibody that recognizes the distal domain of CD6, a highly glycosylated membrane protein predominantly expressed on lymphocytes.[4] The CD6 co-stimulatory pathway contributes to Th1 activation and differentiation of T-cells, promoting a pro-inflammatory response.[4] It is remarkable that these immunomodulatory effects are not produced by inhibiting ligand binding and inducing T-cell depletion. They are effected by turning receptor binding non-productive either by inhibiting new receptor formation or by stimulating the loss of existing receptors or by causing a blockage followed by internalization or downregulation of the receptors. No significant alteration of the lymphocyte count and thereby any signs of immunosuppression are documented at any dose of itolizumab monotherapy; this safety profile being further corroborated by the lowest (18%) rate of infections in its phase III trials in comparison to other biological agents (ustekinumab, 22–31%; etanercept, 27%, adalimumab, 29% and infliximab, 42%).[2],[5]
The patient's demand for a prompt response and the anticipated delayed effect of itolizumab (consequent to it blocking upstream pathways of T-cell activation) were the reasons we decided to co-administer cyclosporine for the initial fortnight. Subsequently, remission continued during monotherapy with itolizumab, PASI-50 was reached at week 4, PASI-75 at week 10 and PASI-90 at week 12.
Use of itolizumab may provide longer remission and increased safety in patients with severe or erythrodermic psoriasis. However, better evidence generated by larger randomized controlled studies is required to corroborate our results and to standardize the relevant guidelines.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
| 1. |
Mrowietz U, Kragballe K, Reich K, Spuls P, Griffiths CE, Nast A, et al. Definition of treatment goals for moderate to severe psoriasis: A European consensus. Arch Dermatol Res 2011;303:1-10.
[Google Scholar]
|
| 2. |
Krupashankar DS, Dogra S, Kura M, Saraswat A, Budamakuntla L, Sumathy TK, et al. Efficacy and safety of itolizumab, a novel anti-CD6 monoclonal antibody, in patients with moderate to severe chronic plaque psoriasis: Results of a double-blind, randomized, placebo-controlled, phase-III study. J Am Acad Dermatol2014;71:484-92.
[Google Scholar]
|
| 3. |
Rosenbach M, Hsu S, Korman NJ, Lebwohl MG, Young M, Bebo BF Jr, et al. Treatment of erythrodermic psoriasis: From the medical board of the National Psoriasis Foundation. J Am Acad Dermatol 2010;62:655-62.
[Google Scholar]
|
| 4. |
Nair P, Melarkode R, Rajkumar D, Montero E. CD6 synergistic co-stimulation promoting proinflammatory response is modulated without interfering with the activated leucocyte cell adhesion molecule interaction. Clin ExpImmunol 2010;162:116-30.
[Google Scholar]
|
| 5. |
Rodriguez PC, Torres-Moya R, Reyes G, Molinero C, Prada D, Lopez AM, et al. A clinical exploratory study with itolizumab, an anti-CD6 monoclonal antibody, in patients with rheumatoid arthritis. Results Immunol 2012;2:204-11.
[Google Scholar]
|
Fulltext Views
2,738
PDF downloads
2,261





