Translate this page into:
Fibrillar IgA staining – An uncommon finding in dermatitis herpetiformis
Corresponding author: Varsha M. Shetty, Department of Dermatology, Kasturba Medical College, Manipal Academy of Higher Education, Manipal, Karnataka, India. varshams18@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Rao R, Pai K, Kurien G, Shetty VM. Fibrillar IgA staining – An uncommon finding in dermatitis herpetiformis. Indian J Dermatol Venereol Leprol 2021;87:574-6.
Sir,
Dermatitis herpetiformis is an autoimmune subepidermal blistering cutaneous disorder with characteristic clinical, histopathological, and immunopathological findings. It classically presents as symmetric, intensely pruritic eruption composed of grouped papules, vesicles, and urticarial plaques primarily affecting the elbows, knees, shoulder, posterior scalp, buttocks, and sacral area with a chronic and relapsing course. Its characteristic histopathological features include subepidermal clefting with papillary dermal neutrophilic microabscess. Histopathology may be non-specific in almost 37.5% patients while detection of circulating autoantibodies against transglutaminase or endomysium may be helpful in some patients for diagnosis.1 Direct immunofluorescence microscopy of perilesional skin is considered the gold standard test for diagnosis. It typically shows granular deposits of IgA in the dermal papillae and/ or along the basement membrane zone. Occasionally, the patients may exhibit a distinct “fibrillar pattern of IgA deposition” on direct immunofluorescence.1,2 Herein, we report three cases of dermatitis herpetiformis showing fibrillar deposits of IgA.
Case 1: A 52-year-old female presented with multiple itchy, vesicular eruptions for 5 months with recent exacerbation for three days. There was no history of gastrointestinal symptoms or mucosal erosions. Cutaneous examination revealed multiple tense, grouped vesicles, and excoriated papules on her upper chest, trunk, abdomen, neck, and extensors of bilateral arms. Dermatitis herpetiformis, vesicular bullous pemphigoid, and transient acantholytic dermatosis were considered as clinical differentials.
Case 2: A 22-year-old female presented with multiple pruritic vesicles on her face, neck, and chest for six years. Her skin problem exacerbated on sun exposure and during summer months. There was no evidence of gastrointestinal symptoms suggestive of gluten sensitivity. We considered hydroa vacciniforme and vesicular bullous pemphigoid as our clinical differentials.
Case 3: A 22-year-old male presented with multiple pruritic vesicles, erosions, and erythematous plaques involving his upper chest, back, and forehead since last four months [Figure 1]. There was no history of gastrointestinal complaints indicative of gluten sensitivity. The clinical differentials considered in this case were dermatitis herpetiformis, linear IgA disease and vesicular bullous pemphigoid.
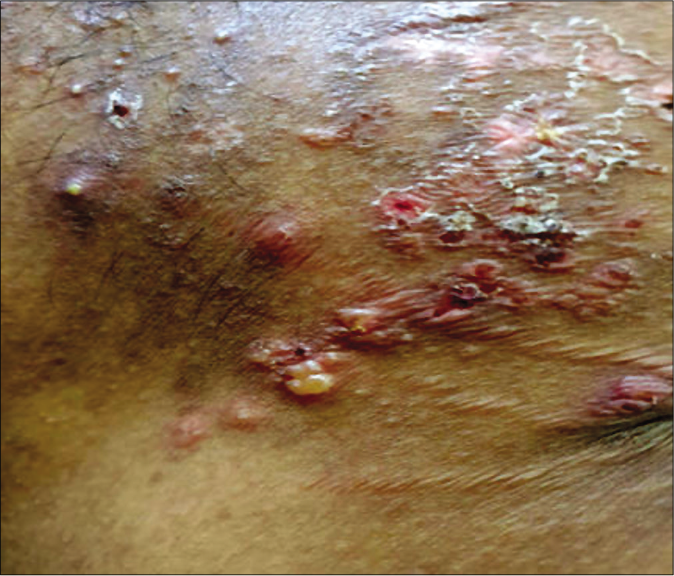
- Case 3 – Grouped vesicles, erythematous plaques, crusting, and scaling distributed on the upper back
The perilesional skin biopsy samples of all cases were sent to the immunofluorescence laboratory, Kasturba Hospital, Manipal, for direct immunofluorescence. The histopathology image could be reviewed in only one case (case three) which revealed a subepidermal blister filled with fibrin, neutrophils, and eosinophils. In addition, eosinophilic spongiosis and perivascular inflammatory infiltrate comprising lymphocytes, histiocytes with occasional neutrophils, and eosinophils were also observed [Figure 2].
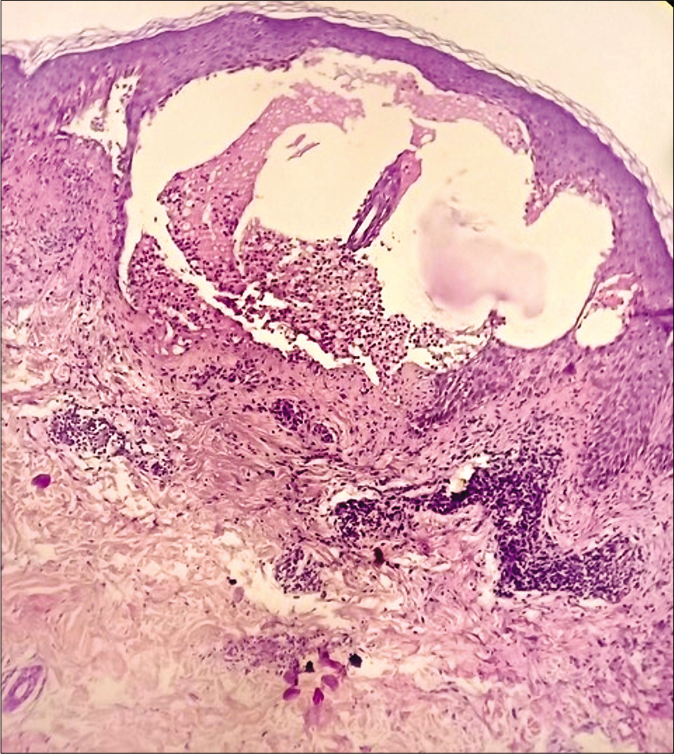
- Case 3 – Subepidermal blister showing collections of predominantly neutrophils with perivascular inflammatory infiltrate of lymphocytes and histiocytes (H and E, ×100)
We prepared 5 μm frozen sections on adhesive slides and stained with fluorescein isothiocyanate-conjugated IgM, IgG, C3, IgA, and fibrinogen as per standard protocol to examine under fluorescence microscope. All three patients exhibited fibrillar pattern of staining with IgA along the dermal papillae and dermoepidermal junction, confirming the diagnosis of dermatitis herpetiformis [Figures 3 and 4]. In addition, a granular C3 staining was seen along the basement membrane zone in case two.
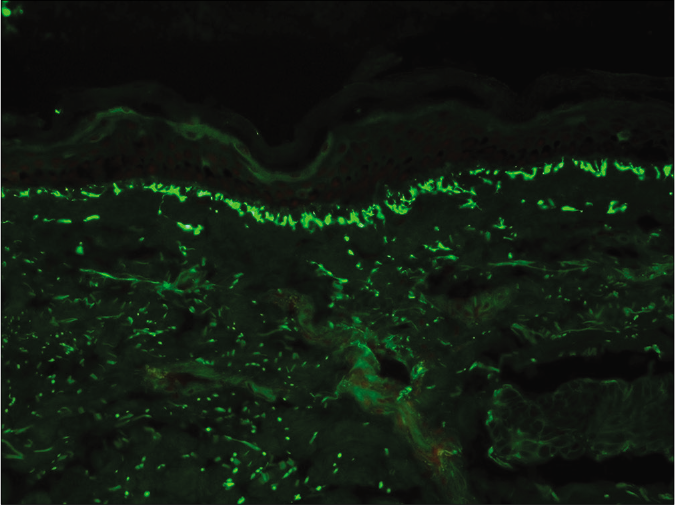
- Case 1 – Fibrillar deposits of IgA seen as linear streaks along the dermal papillae and dermoepidermal junction (fluorescein isothiocyanate, ×200)
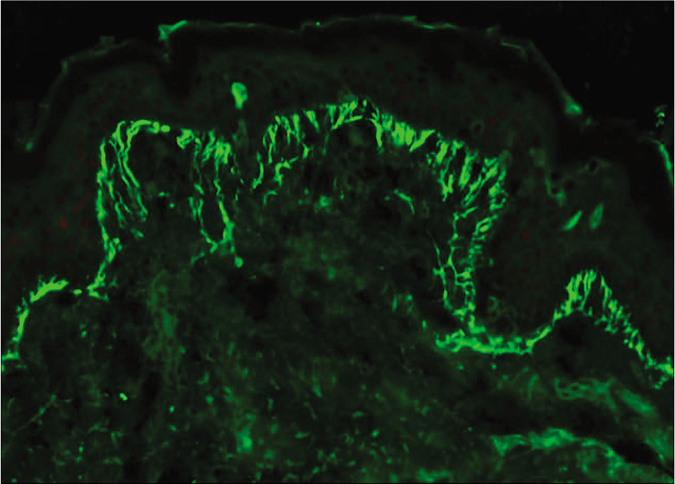
- Case 3 – Fibrillar staining of basement membrane zone and dermal papillae with IgA deposits (fluorescein isothiocyanate, ×400)
Fibrillar pattern of IgA staining in dermatitis herpetiformis was first observed by Kawana and Segawa in 1993.3 The prevalence of fibrillar pattern varies in different ethnic groups; it is encountered less frequently in Caucasians (1.2–2.8 %) compared to Chinese patients (4.5%).4 In contrast, fibrillar pattern is encountered more frequently in Japanese population ranging from 36.3% to 56%.5,6 As direct immunofluorescence is considered the gold standard diagnostic investigation for this condition, it is important to recognize this pattern. The fibrillar pattern is a distinct pattern and not just a mere reflection of longitudinal and transverse sectioning of biopsy. This is corroborated by confocal microscopic studies which demonstrated that the staining pattern continues to be fibrillar irrespective of the type of sectioning. In addition, electron microscopic studies have revealed fibrillar IgA deposits to be diffusely dispersed as fine granules over the entire surface of individual microfibril, in comparison to larger aggregates or clumps of immunoreactants deposited on microfibrils in granular pattern. Further, the location of immune deposits as demonstrated by immunoelectron microscopy is the same in both granular and fibrillar patterns and is centered around the dermal microfibril bundles.3 Occasionally, a combination of granular and fibrillar IgA deposition in the same biopsy specimen has been reported in 5.8% and 7.7% of patients by Ohata et al. and Shibahara et al., respectively.5,6 Miraflor et al. reported a case of dermatitis herpetiformis which showed fibrillar pattern of IgA initially, but a repeat biopsy subsequently revealed a granular IgA deposition raising the possibility of fibrillar pattern being observed during the initial stages of the disease.7 An association between gluten sensitive enteropathy and presence of IgA antibodies to epidermal transglutaminase has been reported in fibrillar dermatitis herpetiformis patients albeit less frequently.8 All these findings support the fact that fibrillar pattern is a distinct variant and not a separate entity. However, classical dermatitis herpetiformis typically involves extensor surfaces of elbows and knees, while these sites are rarely involved in fibrillar type.4 Occasionally, atypical clinical presentations such as urticarial or psoriasiform lesions have been reported.8 Moreover, dapsone is the main stay of treatment and therapeutic response remains same irrespective of the pattern of IgA deposition.6 Japanese patients differ from their Caucasian counterpart in that they have less tendency to develop gluten sensitive enteropathy, regardless of the staining pattern, and are less likely to be associated with HLA DQ2/DQ8 indicating that ethnicity of the patients might play a crucial role in the pathogenesis of this condition.4 To conclude, in the event of atypical clinical features, non-specific histopathology, and negative serology, fibrillar pattern on direct immunofluorescence poses a diagnostic confusion and may mislead the clinician. Hence, it is imperative for dermatologists and dermatopathologists to be aware of fibrillar pattern of IgA deposition in dermatitis herpetiformis.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- Newly described clinical and immunopathological feature of dermatitis herpetiformis. Clin Dev Immunol. 2012;2012:967974.
- [CrossRef] [PubMed] [Google Scholar]
- A case of dermatitis herpetiformis with fibrillar immunoglobulin a deposition: A rare pattern not to be missed. Am J Dermatopathol. 2019;41:511-3.
- [CrossRef] [PubMed] [Google Scholar]
- Confocal laser scanning microscopic and immunoelectron microscopic studies of the anatomical distribution of fibrillar IgA deposits in dermatitis herpetiformis. Arch Dermatol. 1993;129:456-9.
- [CrossRef] [PubMed] [Google Scholar]
- Distinct characteristics in Japanese dermatitis herpetiformis: A review of all 91 Japanese patients over the last 35 years. Clin Dev Immunol. 2012;2012:562168.
- [CrossRef] [PubMed] [Google Scholar]
- Dermatitis herpetiformis in Japan: An update. Dermatology. 2002;204:37-42.
- [CrossRef] [PubMed] [Google Scholar]
- Dermatitis herpetiformis with fibrillar IgA deposition and unusual histologic findings. JAAD Case Rep. 2017;3:344-7.
- [CrossRef] [PubMed] [Google Scholar]
- Fibrillar IgA deposition in dermatitis herpetiformis an underreported pattern with potential clinical significance. J Cutan Pathol. 2010;37:475-7.
- [CrossRef] [PubMed] [Google Scholar]





